Fabrication and Biomedical Applications of Three-Dimensionally Ordered Mesoporous Carbon: A Comprehensive Guide for Drug Development
This article provides a comprehensive overview of the synthesis, functionalization, and application of three-dimensionally ordered mesoporous carbon (3DOMC) materials, with a specialized focus on drug delivery systems.
Fabrication and Biomedical Applications of Three-Dimensionally Ordered Mesoporous Carbon: A Comprehensive Guide for Drug Development
Abstract
This article provides a comprehensive overview of the synthesis, functionalization, and application of three-dimensionally ordered mesoporous carbon (3DOMC) materials, with a specialized focus on drug delivery systems. It explores fundamental principles, including pore architecture and structure-property relationships, before detailing advanced fabrication techniques such as hard-templating and emerging 3D printing methods. The content addresses critical challenges like high production costs and structural stability, offering optimization strategies. Furthermore, it presents rigorous validation through comparative case studies, highlighting the superior performance of 3DOMCs like CMK-8 and CMK-9 in controlled antibiotic and poorly soluble drug release. This resource is tailored for researchers and scientists seeking to leverage these versatile nanomaterials for advanced therapeutic applications.
Unraveling the Architecture and Core Principles of 3D Ordered Mesoporous Carbon
Three-dimensionally ordered mesoporous carbon (3D-OMC) is a class of porous carbon materials characterized by a periodic arrangement of pores with diameters between 2 and 50 nm [1]. These materials are distinguished from disordered porous carbons by their highly uniform, interconnected pore networks, which form a three-dimensional architecture [2] [3]. The first successful synthesis of ordered mesoporous carbon was achieved by Ryoo et al. using mesoporous silica as a hard template, paving the way for targeted design of these materials [1]. In the context of advanced materials research, 3D-OMCs have emerged as crucial components in energy storage, catalysis, and drug delivery systems due to their exceptional structural properties, including high specific surface area, large pore volume, and remarkable thermal and chemical stability [3] [1]. This article defines the fundamental structure and key characteristics of 3D-OMCs, providing application notes and detailed protocols for their fabrication and evaluation.
Structural Definition and Key Characteristics
Fundamental Structural Properties
The defining structural feature of 3D-OMCs is their periodic nanoscale architecture, which consists of a carbon framework arranged in a regular, repeating pattern with uniform pore sizes. According to IUPAC classification, mesopores are defined as having diameters between 2 and 50 nm, and 3D-OMCs exhibit these dimensions in an ordered arrangement [1]. The materials possess several characteristic properties that make them valuable for scientific applications:
- High Specific Surface Area: Typically ranging from 800 to 1500 m²/g, providing numerous active sites for reactions and interactions [3] [1].
- Large Pore Volume: Generally between 1.0-1.5 cm³/g, facilitating efficient mass transport and storage of guest species [4].
- Ordered Pore Architecture: Regular and interconnected pore network enabling precise control over molecular diffusion [2].
- Excellent Electrical Conductivity: Facilitating electron transfer in electrochemical applications [5] [2].
- Thermal and Chemical Stability: Maintaining structural integrity under harsh conditions and in acidic/basic environments [1].
Table 1: Characteristic Structural Parameters of 3D Ordered Mesoporous Carbons
| Parameter | Typical Range | Significance |
|---|---|---|
| Pore Size | 3.9 - 9.4 nm (tunable) [4] | Determinates accessibility for molecules/ions |
| Specific Surface Area | 800 - 1500 m²/g [3] | Provides abundant active sites for reactions/adsorption |
| Pore Volume | 1.0 - 1.5 cm³/g [4] | Influences storage capacity and mass transfer |
| Electrical Conductivity | High (e.g., 4-fold increased over conventional OMC) [2] | Critical for electrochemical applications |
Comparative Structural Advantages
3D-OMCs offer significant advantages over other carbon materials, as summarized in Table 2. Their ordered mesoporous structure provides superior performance characteristics compared to traditional carbon materials.
Table 2: Comparison of 3D-OMC with Other Carbon Materials
| Material Type | Pore Structure | Specific Surface Area (m²/g) | Key Advantages |
|---|---|---|---|
| 3D Ordered Mesoporous Carbon | Ordered, uniform mesopores (3.9-9.4 nm) [4] | 800-1500 [3] | Precise pore control, enhanced mass transfer, excellent conductivity [3] |
| Activated Carbon | Disordered, predominantly micropores | High but microporous [3] | Low cost, high surface area, but limited control and conductivity [3] |
| Biochar | Disordered, broad pore distribution | Lower and rougher [3] | Sustainable source, but irregular pores and lower surface area [3] |
Synthesis Methodologies and Experimental Protocols
The synthesis of 3D-OMCs primarily employs template-based approaches, which provide precise control over the resulting pore structure and architecture. The following sections detail the primary synthesis methodologies.
Hard Template Method
The hard template method, also known as nanocasting, is a widely used approach for synthesizing 3D-OMCs with highly ordered pore structures [3]. This method utilizes a rigid solid template, typically mesoporous silica, to direct the formation of the carbon architecture.
Detailed Experimental Protocol:
- Template Synthesis: Prepare cubic Ia3d mesoporous KIT-6 silica template according to established procedures [4]. Combine 5g Pluronic P123 triblock copolymer with 180g distilled water and 9.9g HCl (35 wt%) with vigorous stirring at 35°C. Add 5g n-butanol, followed by 10.75g tetraethyl orthosilicate (TEOS). Stir continuously for 24h at 35°C, then transfer to autoclave for hydrothermal treatment at 100°C for 24h. Recover solid product by filtration, dry overnight at 100°C, and calcine at 550°C for 6h in air [4].
Precursor Infiltration: Prepare carbon precursor solution by dissolving 0.625g sucrose (95 wt%) and 0.071g sulfuric acid (98 wt%) in 2.5g distilled water. For pore expansion, add 0.113g boric acid (99.5 wt%) as pore expanding agent [4]. Add 0.5g KIT-6 silica template to the solution, then heat at 100°C for 6h, followed by further heating at 160°C for 6h.
Secondary Infiltration (Optional): Repeat the infiltration process using a solution of 0.413g sucrose, 0.047g sulfuric acid, and 0.075g boric acid in 2.5g distilled water to ensure complete pore filling [4].
Carbonization: Place the template-precursor composite in a tube furnace and heat to 900°C under N₂ flow (typically 3h holding time) to convert the organic precursor to carbon [4].
Template Removal: Remove the silica template by washing with 5 wt% HF solution at room temperature, or alternatively with concentrated NaOH solution [4] [1]. Recover the resulting 3D-OMC by filtration, and wash thoroughly with distilled water and ethanol.
Key Advantages and Limitations:
- Advantages: Produces highly ordered structures with excellent pore size control; enables replication of complex template architectures.
- Limitations: Involves multiple steps including template synthesis and removal; uses corrosive HF for silica etching; relatively time-consuming and costly [3].
Soft Template Method
The soft template method utilizes self-assembling block copolymers to direct the formation of mesoporous structures in a single-step process [3].
Detailed Experimental Protocol:
- Solution Preparation: Dissolve amphiphilic block copolymer (typically Pluronic F127) in ethanol or water [3] [6].
Precursor Addition: Add carbon precursor (e.g., phenolic resol) to the polymer solution with stirring to form a homogeneous mixture [6].
Evaporation-Induced Self-Assembly: Cast the solution and allow solvent evaporation at room temperature to facilitate self-assembly of the mesophase [6].
Thermal Treatment: First, heat at 100°C for 24h to crosslink the resin, then carbonize at higher temperatures (350-900°C) under inert atmosphere to convert to carbon and remove the polymer template [6].
Key Advantages and Limitations:
- Advantages: Simpler one-pot synthesis; no template removal step required; more scalable and cost-effective [3].
- Limitations: Lower structural order compared to hard-templated materials; limited to specific precursor-template combinations [3].
Advanced Applications and Performance
Electrochemical Energy Storage and Conversion
3D-OMCs demonstrate exceptional performance in electrochemical applications due to their combination of high surface area, ordered pore structure, and excellent electrical conductivity.
Application Note: Hydrogen Evolution Reaction (HER) Electrocatalyst
- Material System: 1T-phase MoS₂@CMK-3 heterostructure [5]
- Preparation: 1T-phase MoS₂ nanosheets (approximately 80% metallic phase) integrated with 3D ordered mesoporous CMK-3 carbon [5]
- Key Performance Metrics:
- Low Tafel slope of 65 mV dec⁻¹
- Overpotential of 260 mV to reach current density of 10 mA cm⁻²
- Excellent durability in prolonged electrolysis tests
- Enhanced active sites, stability, and diffusion properties
- Advantage: The 3D-OMC support prevents restacking of MoS₂ nanosheets, maintains structural stability, and facilitates efficient electron/ion transport [5]
Microwave Absorption Materials
3D-OMCs functionalized with metal oxides demonstrate exceptional electromagnetic wave absorption capabilities.
Application Note: ZnO-Modified OMC Spheres
- Material System: Ultrafine zinc oxide nanoparticles supported on 3D ordered mesoporous carbon spheres (ZnO/OMCS) [7]
- Preparation: Silica inverse opal template infiltrated with phenolic resol/F127 solution, carbonized at 900°C, then modified with ZnO nanoparticles via sol-gel method [7]
- Key Performance Metrics:
- Strong absorption (-39.3 dB at 10.4 GHz)
- Broad effective absorption bandwidth (9.1 GHz)
- Small thickness requirement (2 mm)
- Advantage: 3D ordered structure promotes multiple reflection and scattering of incident microwaves, while well-dispersed ZnO nanoparticles improve interfacial polarization and impedance matching [7]
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Reagents for 3D-OMC Synthesis via Hard Template Method
| Reagent | Function | Typical Specification | Alternative Options |
|---|---|---|---|
| Pluronic P123 (EO₂₀PO₇₀EO₂₀) | Structure-directing agent for silica template | MW ~5,800 [4] | F127, other amphiphilic block copolymers |
| Tetraethyl Orthosilicate (TEOS) | Silica source for template synthesis | >98% purity [4] | Tetramethyl orthosilicate (TMOS) |
| Sucrose | Carbon precursor | >95% purity [4] | Phenolic resol, furfuryl alcohol |
| Boric Acid (H₃BO₃) | Pore expanding agent | >99.5% purity [4] | Not required for standard synthesis |
| Hydrofluoric Acid (HF) | Silica template removal | 48% solution [4] | Concentrated NaOH for alternative etching |
| Sulfuric Acid (H₂SO₄) | Catalyst for carbon precursor polymerization | 98% concentration [4] | HCl, other acid catalysts |
Structural Characterization Workflow
Comprehensive characterization of 3D-OMCs requires multiple analytical techniques to confirm the ordered structure and determine key physicochemical properties.
Characterization Protocol Details:
X-Ray Diffraction (XRD):
Nitrogen Physisorption:
Electron Microscopy:
Thermal Analysis:
- Purpose: Determine thermal stability and compositional characteristics
- Parameters: TGA under air or N₂ atmosphere, heating rate 5-10°C/min
- Expected Results: High thermal stability with major decomposition above 400°C [5]
Three-dimensionally ordered mesoporous carbons represent a significant advancement in porous material science, offering precisely controlled nanoscale architectures that enable superior performance in diverse applications. Their defining structural characteristics—high surface area, tunable pore sizes, three-dimensional ordering, and excellent electrical conductivity—make them particularly valuable for electrochemical energy systems, catalytic supports, and advanced functional materials. The synthesis methodologies, particularly the hard template approach, provide researchers with robust protocols for fabricating these materials with specific structural parameters. As research in this field continues to evolve, 3D-OMCs are poised to play an increasingly important role in addressing challenges in energy storage, conversion, and advanced materials design through their unique combination of structural precision and functional versatility.
Within the expanding field of carbon materials, three-dimensionally ordered mesoporous carbon (3DOMC) represents a significant architectural advancement compared to conventional carbon workhorses like activated carbon and biochar. While all three are carbon-based, their distinct structural properties, stemming from different fabrication philosophies, dictate their performance in advanced applications. Activated carbon is prized for its high microporosity and extensive surface area, making it a ubiquitous adsorbent [8]. Biochar, often less processed, is recognized for its role in carbon sequestration and soil amendment, while also finding use in environmental remediation [9] [10]. In contrast, 3DOMC is engineered with a precise, ordered pore network that facilitates superior mass transport and exposes a highly accessible surface area, making it particularly suited for high-performance applications in electrocatalysis, energy storage, and drug delivery [3] [11]. This analysis provides a detailed comparison of these materials, supported by quantitative data, experimental protocols, and visualization to guide researchers in selecting and applying the appropriate carbon material for their specific needs.
Material Properties and Comparative Analysis
The distinct value propositions of 3DOMC, activated carbon, and biochar are rooted in their fundamental physical and chemical properties. The table below provides a quantitative comparison of these key characteristics.
Table 1: Comparative properties of 3DOMC, Activated Carbon, and Biochar.
| Property | 3DOMC | Activated Carbon | Biochar |
|---|---|---|---|
| Porosity Type | Ordered mesopores (2-50 nm) [3] | Primarily micropores (<2 nm) [3] | Disordered mix of micro-, meso-, and macropores [3] |
| Structural Order | Highly ordered, periodic structure [11] | Amorphous, disordered network | Amorphous, rough structure [3] |
| Typical Specific Surface Area (m²/g) | High (can exceed 1000) [3] | Very High (can exceed 1500) [3] | Low to Moderate (often < 500) [12] [3] |
| Ion Exchange Capacity | Low | Low [8] | Significant [8] |
| Primary Applications | Electrocatalysis, energy storage, drug delivery [3] [11] | Water/air purification, solvent recovery [8] | Soil amendment, carbon sequestration, remediation [9] [8] |
| Production Energy (MJ/kg) | N/A | 44 - 170 [10] | 1.1 - 16 [10] |
| GHG Emissions (kg CO₂eq/kg) | N/A | 1.2 - 11 [10] | -0.1 to -3.5 (carbon-negative) [10] |
The data reveals a clear trade-off between structural precision and environmental impact. Activated carbon boasts the highest surface area but is energy-intensive to produce. Biochar requires significantly less energy and is carbon-negative, but has a lower and less consistent surface area [10]. 3DOMC occupies a specialized niche, where its ordered mesoporosity is not about maximizing raw surface area, but about optimizing accessibility and transport for demanding applications like fast ion transfer in batteries or the immobilization of large biomolecules [3].
Synthesis and Fabrication Protocols
Synthesis of Three-Dimensionally Ordered Mesoporous Carbon (3DOMC)
The synthesis of 3DOMC typically relies on a nanocasting technique using a hard template to create the ordered porous network. The following protocol, adapted from recent literature, details the synthesis of a 3DOMC scaffold suitable for supporting single-atom catalysts [7] [11].
Title: Hard-Template Synthesis of 3DOMC via Superlattice Blotting
Research Reagent Solutions:
- Template: Poly(methyl methacrylate) (PMMA) colloidal crystal or silica inverse opal.
- Carbon Precursor: Phenolic resol (Mw < 500).
- Structure Director: Triblock copolymer F127 (EO₁₀₆PO₇₀EO₁₀₆).
- Solvents: Ethanol, Methanol.
- Etching Agent: Hydrofluoric Acid (HF, 5%) or Potassium Hydroxide (KOH) solution.
Experimental Workflow:
Detailed Protocol:
- PMMA Colloidal Crystal Template: Self-assemble monodisperse PMMA spheres into a close-packed array to create a three-dimensional template.
- Silica Inverse Opal Fabrication: a. Prepare a silica precursor solution by mixing tetraethoxysilane (TEOS), 0.1 M HCl, and ethanol (mass ratio 1:1:1.5). Stir for 1 hour [7]. b. Immerse the PMMA template in the silica precursor solution. Allow it to soak for 1 hour. c. Remove the template and dry it at room temperature. d. Calcinate the composite at 450°C for 5 hours to remove the PMMA template, resulting in a silica inverse opal structure [7].
- Carbon Source Infiltration: a. Impregnate the silica inverse opal template with an ethanol solution containing resol and F127. b. Slowly evaporate the ethanol at room temperature to allow the precursors to fill the template's pores. c. Thermo-polymerize the infiltrated precursor by heating at 100°C for 24 hours, followed by a stepwise carbonization under nitrogen (e.g., 350°C for 5 hours to remove F127, then 900°C for 2 hours for carbonization) [7].
- Template Removal: Etch away the silica framework by immersing the carbon/silica composite in a 5% HF solution for several days.
- Post-functionalization (Optional): The resulting 3DOMC can be modified with metal atoms (e.g., Ni) or heteroatoms (e.g., N, S, P) via impregnation and a second high-temperature treatment to create active sites for catalysis [11].
Production of Activated Carbon from Biomass
Activated carbon is commonly produced from biomass precursors like corn stover through a two-step process involving slow pyrolysis followed by chemical activation [12].
Title: Two-Step Synthesis of Activated Carbon via KOH Activation
Research Reagent Solutions:
- Feedstock: Dried and milled corn stover (or other lignocellulosic biomass).
- Activating Agent: Potassium Hydroxide (KOH) pellets.
- Atmosphere: Inert gas (N₂ or Ar).
Experimental Workflow:
Detailed Protocol:
- Pyrolysis: Subject the dried biomass to slow pyrolysis in an inert atmosphere. Use a moderate temperature (e.g., below 500°C) and a slow heating rate (e.g., 10°C/min) to produce a biochar precursor [12].
- Chemical Activation: a. Mix the biochar with KOH at a specified mass ratio (e.g., 1:1 to 1:4 KOH:biochar) [12]. b. Heat the mixture under an inert atmosphere to a high temperature (e.g., 700-800°C) for 1-2 hours. The KOH reacts with carbon to create metallic K, K₂O, and K₂CO₃, which intercalate and etch the carbon matrix, generating extensive microporosity [12].
- Post-processing: After cooling, wash the resulting activated carbon with dilute acid and copious deionized water to remove residual chemicals and soluble salts. Dry the product overnight at 105°C [12].
Production of Biochar via Hydrothermal Carbonization (HTC)
Biochar can be produced through various methods, with HTC being a "greener" alternative to pyrolysis as it uses hot, compressed water [12].
Title: Biochar Production via Hydrothermal Carbonization
Research Reagent Solutions:
- Feedstock: Wet biomass (e.g., corn stover, no drying required).
- Solvent: Deionized water.
- Reactor: Sealed hydrothermal reactor (autoclave).
Detailed Protocol:
- Reactor Loading: Load the biomass and water into a sealed hydrothermal reactor.
- HTC Reaction: Heat the reactor to a target temperature between 180°C and 250°C [12]. Maintain the temperature (dwell time) for a set period, typically 1 to 4 hours [12].
- Product Separation: After the reaction, allow the reactor to cool. Separate the solid fraction (hydrochar) from the liquid bio-oil by filtration.
- Drying: Dry the resulting hydrochar at 105°C to produce the final biochar. The surface area of HTC biochar is generally low but can be increased with higher temperatures and dwell times, as the process breaks down the cellulosic and hemicellulosic components of the biomass [12].
Application Notes
Electrocatalysis
The ordered mesopores of 3DOMC are critical for high-performance electrocatalysis. They stabilize gas-liquid-solid interfaces, facilitate rapid mass transport of electrolytes and gases, and provide a high surface area for dispersing active sites. For instance, a 3DOMC support with Ni single-atom active sites coordinated with N and S (Ni-N₂S₂) demonstrated an overpotential of only 239 mV for the oxygen evolution reaction (OER) at 20 mA cm⁻², outperforming commercial RuO₂ catalysts [11]. The ordered structure prevents bubble accumulation and pore clogging, ensuring stability for over 100 hours [11].
Application Suggestion: Utilize 3DOMC as a catalyst support in electrochemical cells where high current density and long-term stability are required. The synthesis protocol in Section 3.1 is directly applicable.
Environmental Adsorption
Activated carbon remains the benchmark for adsorbing small organic contaminants from air and water due to its enormous microporous surface area. It is highly effective for removing pollutants like phenolic compounds (e.g., vanillin), with studies showing removal efficiencies up to 98% [12]. Biochar, with its lower cost and carbon-negative footprint, is a compelling alternative for certain remediation tasks, particularly where its cation exchange capacity can be leveraged for heavy metal removal [8] [10]. Some biochars can even surpass activated carbon in adsorption capacity for specific metals like cadmium, despite having a much lower surface area [10].
Application Suggestion: For purifying water containing trace organic pharmaceuticals or solvents, use activated carbon. For large-scale, in-situ soil remediation involving mixed contaminants, especially heavy metals, biochar may be more cost-effective and sustainable.
Drug Delivery and Biomedicine
The tunable, ordered mesopores of 3DOMC (2-50 nm) are ideal for loading and controlling the release of therapeutic molecules, proteins, or nucleic acids. Their high surface area and biocompatibility make them excellent candidates for drug delivery systems [3]. The interconnected pore network ensures uniform drug loading and sustained release kinetics.
Application Suggestion: For targeted drug delivery or biosensing, 3DOMC spheres can be functionalized with specific ligands. Their pore size can be tailored during synthesis to match the hydrodynamic diameter of the drug molecule for optimal loading and release.
The Scientist's Toolkit
Table 2: Essential research reagents for fabricating 3DOMC materials.
| Reagent / Material | Function in Synthesis | Exemplary Use Case |
|---|---|---|
| PMMA or Silica Nano-spheres | Serves as a hard template to create the ordered 3D macroporous or mesoporous structure. | Creating the initial inverse opal scaffold [7] [11]. |
| Phenolic Resol | Polymerizable carbon precursor that infiltrates the template and forms a continuous carbon framework upon pyrolysis. | Source of conductive carbon matrix [7]. |
| Triblock Copolymer (e.g., F127) | Acts as a soft template within the hard template to create mesopores in the carbon walls, adding a second level of porosity. | Generating mesoporosity in the carbon walls of the 3DOMC structure [7]. |
| Hydrofluoric Acid (HF) | Highly effective etchant for removing silica-based hard templates after carbonization. | Final liberation of the 3DOMC structure from the silica inverse opal [7]. |
| Heteroatom Precursors (e.g., Urea, Phosphines) | Source of dopant atoms (N, P, S) that modify the electronic structure of carbon, enhancing catalytic activity. | Creating Ni-N₃P active sites for the hydrogen evolution reaction (HER) [11]. |
| Metal Salts (e.g., Ni Acetate) | Precursor for creating single-atom or nanoparticle metal active sites supported on the 3DOMC. | Dispersing single-atom nickel catalysts for electrocatalysis [11]. |
Ordered mesoporous carbons (OMCs) with three-dimensional (3D) pore architectures represent a significant advancement in nanostructured materials. Defined by their pore sizes between 2-50 nm, these materials possess high specific surface areas, large pore volumes, and interconnected networks that facilitate efficient mass transport [13] [14]. Among these, CMK-8 and CMK-9 exhibit particularly sophisticated gyroidal structures with cubic symmetry, making them exceptional candidates for applications requiring intricate molecular pathways and accessibility [15]. The international union of pure and applied chemistry (IUPAC) classification system categorizes these materials as mesoporous, distinguishing them from microporous (<2 nm) and macroporous (>50 nm) materials [13] [16].
The transition from two-dimensional to three-dimensional pore geometries in mesoporous carbons has unlocked new possibilities in nanomaterial design. While 2D hexagonal structures like CMK-3 (replicated from SBA-15 silica templates) provide well-defined channels, 3D architectures offer enhanced interconnectivity that minimizes diffusion limitations and increases accessibility to internal surfaces [17]. This structural advantage proves critical in applications such as electrocatalysis, where oxygen diffusion pathways directly influence reaction efficiency, and drug delivery, where controlled release profiles depend on pore interconnectivity [18] [17]. The geometric properties of these nanoscale environments directly govern their performance in technological applications, making pore geometry a fundamental consideration in materials design.
Structural Characteristics of 3D Mesoporous Carbon Platforms
CMK-8 and CMK-9 Gyroidal Architectures
CMK-8 and CMK-9 carbon materials exhibit highly symmetric gyroidal structures at the nanometer scale, corresponding to regular, continuous nanopore systems with cubic symmetry [15]. These structures are amorphous at the atomic length scale but demonstrate remarkable long-range order in their pore arrangement. The gyroidal structure forms a continuous network that minimizes diffusion barriers while maximizing surface area accessibility, creating an optimal environment for processes requiring efficient mass transport.
CMK-8 is synthesized using KIT-6 silica as a hard template, which possesses a cubic Ia3d symmetry [17]. This template structure results in a carbon replica with interconnected mesopores that form a 3D network. The replication process creates a carbon framework that inversely mirrors the porous structure of the KIT-6 template, resulting in a material with uniform pore size distribution and excellent structural stability.
CMK-9 features a more complex bimodal porosity within its gyroidal structure [15]. This unique characteristic provides two distinct pore size distributions within the same material, creating a hierarchical system that enhances molecular accessibility. The volume fraction of carbon versus pore volume (effectively the "pore wall thickness") significantly impacts the relative diffraction peak intensities in X-ray characterization, suggesting that careful evaluation of experimental low-angle XRD patterns offers detailed information about nanostructural properties beyond mere geometry identification [15].
Comparative Analysis of 3D Mesoporous Carbon Geometries
Table 1: Structural Parameters and Properties of 3D Ordered Mesoporous Carbons
| Material | Space Group | Template | Pore Structure | Specific Surface Area | Primary Applications |
|---|---|---|---|---|---|
| CMK-8 | Cubic Ia3d | KIT-6 silica | 3D interconnected network | High (>1000 m²/g) | Electrocatalysis, Adsorption |
| CMK-9 | Cubic gyroidal | - | Bimodal porosity | Varies with carbon fraction | Materials with size-selective accessibility |
| CMK-3 | 2D hexagonal (p6mm) | SBA-15 silica | 2D channel array | >2000 m²/g | Drug delivery, Catalysis |
| CMK-3/8 | Composite | Dual SBA-15/KIT-6 | Integrated micro/mesoporosity | Lower than single templates | Balanced ORR activity/stability |
Table 2: Performance Comparison of CMK Materials in Oxygen Reduction Reaction (ORR)
| Electrocatalyst | Onset Potential (Alkaline) | Onset Potential (Acid) | Stability | ORR Pathway | Mass Transport Efficiency |
|---|---|---|---|---|---|
| Fe-N@CMK-3 | 0.99 V~RHE~ | 0.82 V~RHE~ | Best in alkaline | Quasi-4e⁻, shifts slightly | Excellent |
| Fe-N@CMK-8 | Lower than CMK-3 | Lower than CMK-3 | Superior retention in acid | Shifts to 2e⁻ pathway | Limited by microporosity |
| Fe-N@CMK-3/8 | Intermediate | Intermediate | Medium in both media | Sustains 4e⁻ pathway | Balanced |
The structural differences between these mesoporous carbon platforms directly influence their functional performance. CMK-3's 2D hexagonal pores facilitate efficient oxygen diffusion, resulting in superior ORR activity [17]. In contrast, CMK-8's microporous network exhibits lower ORR activity due to limited oxygen accessibility, despite its 3D interconnectivity [17]. This limitation arises from mass transport constraints caused by water flooding in micropores, which restricts reactant access to active sites.
The dual-templated Fe-N@CMK-3/8 represents an engineering solution that bridges the structural properties of both materials, combining micro/mesoporosity to deliver balanced performance [17]. This hierarchical approach demonstrates how pore architecture can be tailored to optimize specific functional characteristics, creating materials with customized properties for targeted applications.
Synthesis Protocols and Methodologies
Hard-Template Synthesis of CMK-8
The synthesis of CMK-8 follows a nanocasting approach using KIT-6 mesoporous silica as a hard template. This method involves multiple precise steps to ensure faithful replication of the 3D cubic structure.
Protocol: CMK-8 Synthesis via KIT-6 Templating
- Template Preparation: Synthesize KIT-6 silica template according to established procedures [17]. The synthesis relies on soft-templating using Pluronic P123 [(PEO)~20~-(PPO)~70~-(PEO)~20~] as a structure-directing agent. The addition of cosurfactants (e.g., butanol) swells the PPO units, reducing micellar interfacial curvature and inducing a transition from hexagonal to cubic symmetry characteristic of KIT-6.
- Precursor Infiltration: Prepare a carbon precursor solution containing 1,10-phenanthroline as nitrogen/carbon source and iron(III) nitrate nonahydrate as metal precursor [17]. Use incipient wetness impregnation to ensure complete pore-filling of the KIT-6 template. The precursor solution concentration should be optimized to achieve complete pore filling without excess external deposition.
- Carbonization: Transfer the impregnated template to a tubular furnace and heat under inert atmosphere (nitrogen or argon) using a controlled temperature program:
- Ramp from room temperature to 350°C at 1°C/min
- Hold at 350°C for 4 hours for stabilization
- Ramp to 800-900°C at 5°C/min
- Maintain at final temperature for 2 hours for complete carbonization
- Template Removal: After cooling to room temperature, remove the silica template by etching with hydrofluoric acid (5-10% v/v) or strong alkaline solution (e.g., 5M NaOH) at room temperature for 24 hours. Recover the CMK-8 product by filtration, wash thoroughly with deionized water and ethanol, and dry under vacuum at 120°C for 12 hours.
Quality Control: Confirm successful replication by powder X-ray diffraction (PXRD), which should show characteristic patterns consistent with the cubic Ia3d structure [17] [15]. Nitrogen physisorption should reveal type-IV isotherms with specific surface area exceeding 1000 m²/g.
Direct Synthesis from Thermoplastic Elastomers
Recent advances have demonstrated alternative pathways to ordered mesoporous carbons through direct pyrolysis of thermoplastic elastomers (TPEs), offering a potentially more scalable approach to 3D nanostructured carbons.
Protocol: Direct Conversion of SEBS to Ordered Mesoporous Carbon
- Material Preparation: Use commercially available polystyrene-block-poly(ethylene-ran-butylene)-block-polystyrene (SEBS) as precursor [19]. Subject the polymer to thermal annealing at 160°C for 12 hours to achieve long-range ordering in their nanostructures.
- Sulfonation-Based Crosslinking: Submerge the annealed SEBS in concentrated sulfuric acid and react at 150°C for 4 hours to selectively crosslink the olefinic block (PEB) [19]. Monitor reaction progress through mass gain and gel fraction, which typically plateau after approximately 4 hours.
- Thermal Treatment: After sulfonation, calcinate the crosslinked polymer under inert atmosphere to selectively decompose the PS minority phase, generating mesopores [19]. For complete carbonization, expose to temperatures above 600°C to convert the PEB matrix to a carbon framework.
This method produces large-pore, heteroatom-doped OMCs with sulfur incorporation from the sulfuric acid treatment [19]. The resulting materials exhibit pore textures tailorable by varying precursor identities and processing parameters.
Application-Specific Performance and Experimental Protocols
Electrocatalysis: Oxygen Reduction Reaction (ORR) Protocol
The oxygen reduction reaction is crucial for fuel cell technologies, and pore geometry significantly influences ORR efficiency. The following protocol evaluates CMK-based electrocatalysts for ORR application.
Experimental Protocol: ORR Performance Assessment
Electrode Preparation:
- Prepare catalyst ink by dispersing 5 mg of CMK material in 1 mL of ethanol with 20 μL Nafion solution (5 wt%)
- Sonicate for 30 minutes to form homogeneous suspension
- Deposit 10 μL of ink onto glassy carbon electrode (diameter: 5 mm)
- Dry at room temperature to form uniform catalyst layer
Electrochemical Measurements:
- Use standard three-electrode cell with catalyst-coated glassy carbon as working electrode
- Employ platinum wire as counter electrode and Ag/AgCl as reference electrode
- Perform cyclic voltammetry in nitrogen-saturated 0.1 M KOH electrolyte
- Conduct linear sweep voltammetry in oxygen-saturated electrolyte at rotating speeds from 400 to 1600 rpm
- Record data at scan rate of 10 mV/s
Data Analysis:
- Calculate electron transfer number using Koutecky-Levich equation
- Determine onset potential and half-wave potential from ORR polarization curves
- Assess stability through accelerated durability tests (3000 potential cycles)
Expected Outcomes: Fe-N@CMK-3 typically demonstrates superior ORR activity with onset potential of 0.99 V~RHE~ in alkaline media, while Fe-N@CMK-8 shows lower activity due to mass transport limitations despite its 3D structure [17]. The dual-templated Fe-N@CMK-3/8 exhibits balanced performance, sustaining a 4e⁻ pathway with medium stability in both acid and alkaline media [17].
Drug Delivery: Bioactive Molecule Loading and Release Protocol
Mesoporous carbon nanoparticles serve as efficient carriers for therapeutic compounds, with pore geometry influencing loading capacity and release kinetics.
Experimental Protocol: Drug Loading and Release Profiling
Material Functionalization:
- Oxidize MCNs using concentrated H~2~SO~4~/HNO~3~ (3:1 v/v) at 60°C for 6 hours to create carboxyl groups [20]
- Alternatively, use ammonium persulfate in dilute H~2~SO~4~ for gentler oxidation [20]
- Purify by centrifugation and redispersion in deionized water
- Characterize surface functionality by FTIR and zeta potential measurements
Drug Loading:
- Select appropriate loading method based on drug properties:
- Solvent evaporation: Dissolve drug and MCNs in organic solvent, evaporate under reduced pressure
- Adsorption equilibrium: Soak MCNs in drug solution, stir for 24 hours, collect by centrifugation
- Melting method: Heat drug above melting point with MCNs, cool slowly
- Determine loading efficiency by measuring supernatant concentration via HPLC or UV-Vis
- Select appropriate loading method based on drug properties:
Release Kinetics:
- Place drug-loaded MCNs in dialysis membrane (MWCO: 12-14 kDa)
- Immerse in release medium (PBS, pH 7.4) at 37°C with continuous shaking
- Collect aliquots at predetermined time intervals
- Analyze drug concentration by validated analytical method
- Fit release data to mathematical models (zero-order, first-order, Higuchi, Korsmeyer-Peppas)
Application Notes: CMK-3 type materials with 2D hexagonal pores demonstrate excellent drug loading capacity, with reported efficiency up to 73.6% for carvedilol [20]. The pore size and surface chemistry significantly influence release profiles, with thicker mesoporous shells decelerating dissolution rates [20]. For cancer therapy applications, folate or hyaluronic acid modification enables targeted delivery to tumor cells [14].
Table 3: Research Reagent Solutions for Mesoporous Carbon Synthesis and Application
| Reagent/Category | Function/Purpose | Application Context | Representative Examples |
|---|---|---|---|
| Structure-Directing Agents | Template formation for pore structure | Hard template synthesis | Pluronic P123, F127, KIT-6 silica, SBA-15 silica |
| Carbon Precursors | Source of carbon framework | Carbonization process | Sucrose, phenolic resin, 1,10-phenanthroline, furfuryl alcohol |
| Dopant Precursors | Introduce heteroatoms for functionality | Electronic property modification | Iron(III) nitrate, ammonium persulfate, dicyandiamide, urea |
| Surface Modifiers | Enhance hydrophilicity/biocompatibility | Drug delivery systems | HNO~3~/H~2~SO~4~, polyethylene glycol (PEG), folate, hyaluronic acid |
| Therapeutic Agents | Payload for delivery systems | Biomedical applications | Doxorubicin, carvedilol, celecoxib, mitoxantrone, ruthenium dyes |
The strategic importance of pore geometry in CMK-8, CMK-9, and other 3D mesoporous carbon structures extends across multiple disciplines, from energy conversion to biomedical applications. The gyroidal architectures of CMK-8 and CMK-9 provide unique 3D interconnected pathways that enable efficient mass transport while maintaining high surface area accessibility [17] [15]. These structural advantages manifest in enhanced performance metrics, whether in electrocatalytic activity, drug loading capacity, or molecular separation efficiency.
Future research directions should focus on precision engineering of pore architectures to create optimized environments for specific applications. The development of dual-templating approaches that combine complementary pore structures represents a promising strategy for achieving balanced performance characteristics [17]. Additionally, the exploration of sustainable synthesis routes using biomass-derived precursors or scalable polymer templates may address current limitations in production cost and environmental impact [19] [16]. As characterization techniques advance, particularly in situ methods for probing pore functionality under operational conditions, our understanding of structure-property relationships will continue to refine, enabling the rational design of next-generation mesoporous carbon materials with tailored geometries for specialized applications.
The development of three-dimensionally ordered mesoporous carbon (3DOMC) materials represents a significant advancement in nanomaterials research, particularly for biomedical applications. These materials are defined by their highly ordered pore networks with tunable diameters between 2 and 50 nm, which confer exceptional physicochemical properties including unprecedented specific surface area, substantial pore volume, and demonstrated biocompatibility [16]. The integration of these three fundamental properties—high surface area, large pore volume, and biocompatibility—enables 3DOMCs to function as superior platforms for drug delivery, surpassing the capabilities of traditional nanocarriers [20] [18]. This document outlines the essential material properties, quantitative performance metrics, and detailed experimental protocols for the fabrication and application of 3DOMCs in drug delivery systems, providing researchers with practical guidance for leveraging these advanced materials.
Essential Properties and Quantitative Metrics
The performance of 3DOMCs in drug delivery applications is governed by a suite of interconnected physicochemical properties. High specific surface area provides extensive interfaces for drug molecule attachment, while large pore volume accommodates substantial therapeutic payloads. The ordered mesoporous structure ensures uniform and predictable drug loading and release kinetics. Furthermore, biocompatibility is a critical requirement for any material intended for biomedical use, ensuring minimal adverse effects when introduced to biological systems [20] [16].
Table 1: Quantitative Properties of Representative Mesoporous Carbon Materials
| Material | Specific Surface Area (m²/g) | Pore Volume (cm³/g) | Primary Pore Size (nm) | Key Application |
|---|---|---|---|---|
| Longan Seed Mesoporous Carbon [21] | 1773 | 1.075 (total) | Mesoporous range | Methylene blue adsorption (1000 mg/g capacity) |
| CMK-9 Ordered Mesoporous Carbon [22] | 1130 | Not specified | Dual mesoporosity | Cephalexin drug delivery (354 mg/g capacity) |
| N-doped Hollow Carbon Nanocapsules (NHCNC-3) [23] | 1400 | Not specified | Hierarchical porosity | Supercapacitors & biocompatible carrier |
| Hollow Mesoporous Carbon (HMC) [20] | Not specified | Not specified | Mesoporous range | Carvedilol delivery (73.6% loading efficiency) |
Table 2: Biocompatibility Assessment of Mesoporous Carbon Materials
| Material | Test Method | Key Findings | Citation |
|---|---|---|---|
| N-doped Hollow Carbon Nanocapsules (NHCNCs) | Cytotoxicity assay with human cells | Negligible cytotoxicity observed, confirming high biocompatibility | [23] |
| Mesoporous Carbon for Colon Cancer Treatment | In vitro and in vivo toxicology evaluation | Showed good biocompatibility in both cytotoxicity and mouse models | [16] |
| Mesoporous Carbon Nanoparticles (MCNs) | Preclinical biodistribution and hemocompatibility | Biocompatible at adequate doses; surface modification enhances biocompatibility | [20] |
Experimental Protocols
Synthesis of Three-Dimensionally Ordered Mesoporous Carbon (3DOMC) via Hard-Templating
The hard-template method, particularly using silica templates, provides precise control over the pore structure and ordering of mesoporous carbons [3] [16].
Materials:
- Template: KIT-6 mesoporous silica or silica inverse opal (from PMMA template) [22] [7].
- Carbon Precursor: Phenolic resol, sucrose, or furfuryl alcohol [22] [7].
- Catalyst: Ethylenediamine (EDA) or sulfuric acid (H₂SO₄) [22] [23].
- Solvents: Ethanol, deionized water.
- Inert Atmosphere: Nitrogen or argon gas.
Procedure:
- Template Preparation: Synthesize or procure the 3D ordered silica template (e.g., KIT-6 for cubic structures or silica inverse opal from PMMA colloidal crystals) [22] [7].
- Precursor Infiltration:
- Prepare a solution containing the carbon precursor (e.g., 1g phenolic resol in 20mL ethanol), and catalyst if required [7].
- Immerse the silica template in the precursor solution to ensure complete infiltration into the mesopores. This can be achieved by stirring or sonication for 1-3 hours [22].
- Evaporate the solvent slowly at room temperature to allow for complete polymerization and filling of the template pores [7].
- Carbonization:
- Place the precursor-filled template in a tube furnace under an inert nitrogen atmosphere.
- Heat to 350°C at 1°C/min and hold for 5 hours to remove any surfactant templates (e.g., F127) [7].
- Increase the temperature to 800-900°C at a rate of 5°C/min and maintain for 2 hours to carbonize the organic precursor into a rigid carbon framework [7].
- Template Removal:
- After cooling to room temperature, immerse the carbon/silica composite in a 5-10% hydrofluoric (HF) acid solution or a concentrated sodium hydroxide (NaOH) solution for 24-72 hours to completely dissolve the silica template [7].
- Collect the resulting 3DOMC by filtration, and wash thoroughly with ethanol and deionized water until neutral pH is achieved.
- Dry the final product in an oven at 60-80°C overnight [7].
Characterization: The synthesized 3DOMC should be characterized using Nitrogen adsorption-desorption isotherms (BET surface area, pore volume), Transmission Electron Microscopy (TEM), and X-ray Diffraction (XRD) to confirm the ordered mesoporous structure and textural properties [21] [23].
Protocol for Drug Loading and In-Vitro Release Studies
This protocol details the process of loading a drug molecule (e.g., an antibiotic) into the 3DOMC and evaluating its release profile under simulated physiological conditions [22].
Materials:
- Mesoporous Carbon Carrier: Synthesized 3DOMC (e.g., CMK-8, CMK-9).
- Drug Molecule: Cephalexin (CFX) or other model drugs (e.g., Methylene Blue for initial studies).
- Solvents: Phosphate Buffered Saline (PBS), ethanol, water.
- Buffer Solutions: PBS at pH 1.2 (simulated gastric fluid) and pH 6.8 (simulated intestinal fluid).
Procedure:
- Surface Functionalization (Optional but Recommended):
- To enhance aqueous dispersion and control drug release, functionalize the 3DOMC surface with amino groups using 3-aminopropyltriethoxysilane (APTES) via a grafting procedure [22].
- Drug Loading:
- Prepare a concentrated solution of the drug (e.g., CFX) in a suitable solvent.
- Dissolve the 3DOMC material in the drug solution at a defined ratio (e.g., 1:10 w/v solid-to-solution ratio).
- Stir the mixture for 24 hours in the dark at room temperature to reach adsorption equilibrium [22].
- Separate the drug-loaded 3DOMC by centrifugation and wash the pellet gently with water to remove surface-adsorbed drug molecules.
- Dry the final drug-loaded carrier in a desiccator under vacuum.
- Calculate the Drug Loading Capacity (DLC) using the formula: DLC (mg/g) = (Weight of loaded drug / Weight of drug-loaded carrier) × 1000 [22].
- In-Vitro Drug Release:
- Place a known quantity of the drug-loaded 3DOMC into release media (e.g., PBS at pH 1.2 and pH 6.8) under constant agitation at 37°C to simulate physiological conditions [22].
- At predetermined time intervals, withdraw a sample of the release medium and replace it with an equal volume of fresh buffer to maintain sink conditions.
- Analyze the drug concentration in the collected samples using a suitable analytical method (e.g., UV-Vis spectroscopy).
- Calculate the cumulative drug release percentage and plot the release profile over time.
Kinetic Modeling: Fit the release data to various kinetic models (e.g., Weibull model, pseudo-second-order) to understand the release mechanism, which is often governed by Fickian diffusion controlled by the material's porosity and electrostatic interactions [22].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for 3DOMC Synthesis and Drug Loading
| Reagent | Function/Role | Example/Citation |
|---|---|---|
| KIT-6 Mesoporous Silica | Hard template for creating ordered 3D mesoporous carbon structures (e.g., CMK-8, CMK-9) | [22] |
| Phenolic Resol | Carbon precursor that infiltrates the template and forms the carbon framework upon pyrolysis | [7] |
| Furfuryl Alcohol | Carbon precursor for synthesizing specific carbon structures like CMK-9 with dual mesoporosity | [22] |
| APTES (3-Aminopropyltriethoxysilane) | Surface functionalization agent to introduce amine groups, improving hydrophilicity and controlled drug release | [22] |
| Hydrofluoric Acid (HF) | Etching agent for the selective removal of the silica template after carbonization | [7] |
| Pluronic F127 | Soft template/surfactant used in combination with hard templates to fine-tune mesoporosity | [7] |
Property Interrelationships and Performance
The exceptional performance of 3DOMCs in drug delivery arises from the synergistic relationship between their key properties. The high surface area, often exceeding 1000 m²/g, provides numerous active sites for drug adsorption, while the large pore volume allows for high drug loading capacities, as demonstrated by the 1000 mg/g uptake of methylene blue [21] and the 354 mg/g loading of cephalexin [22]. The ordered 3D mesoporous structure is crucial for controlling drug release kinetics. It facilitates consistent diffusion pathways, leading to predictable and sustained release profiles, as opposed to the burst release often seen with amorphous carriers. Furthermore, the carbon surface can be readily functionalized (e.g., with APTES) to introduce functional groups that enhance aqueous dispersion, modulate drug-carrier interactions, and enable controlled release in response to specific physiological stimuli like pH changes [22] [20]. Finally, confirmed biocompatibility ensures that these high-performance materials can be safely deployed in biological systems, making them suitable for in-vivo applications [23] [16].
The integration of high surface area, substantial pore volume, and inherent biocompatibility makes three-dimensionally ordered mesoporous carbon a transformative material for advanced drug delivery applications. The structured protocols and data outlined in this document provide a foundation for researchers to synthesize, characterize, and utilize 3DOMCs effectively. Future research directions should focus on refining large-scale production techniques, further exploring the long-term in-vivo biocompatibility and biodistribution of these materials, and designing more sophisticated surface modification strategies to achieve targeted and stimulus-responsive therapeutic delivery. By mastering the essential properties and protocols detailed herein, scientists and drug development professionals can harness the full potential of 3DOMCs to create next-generation nanomedicines.
Structure-Property Relationships in Biomedical Environments
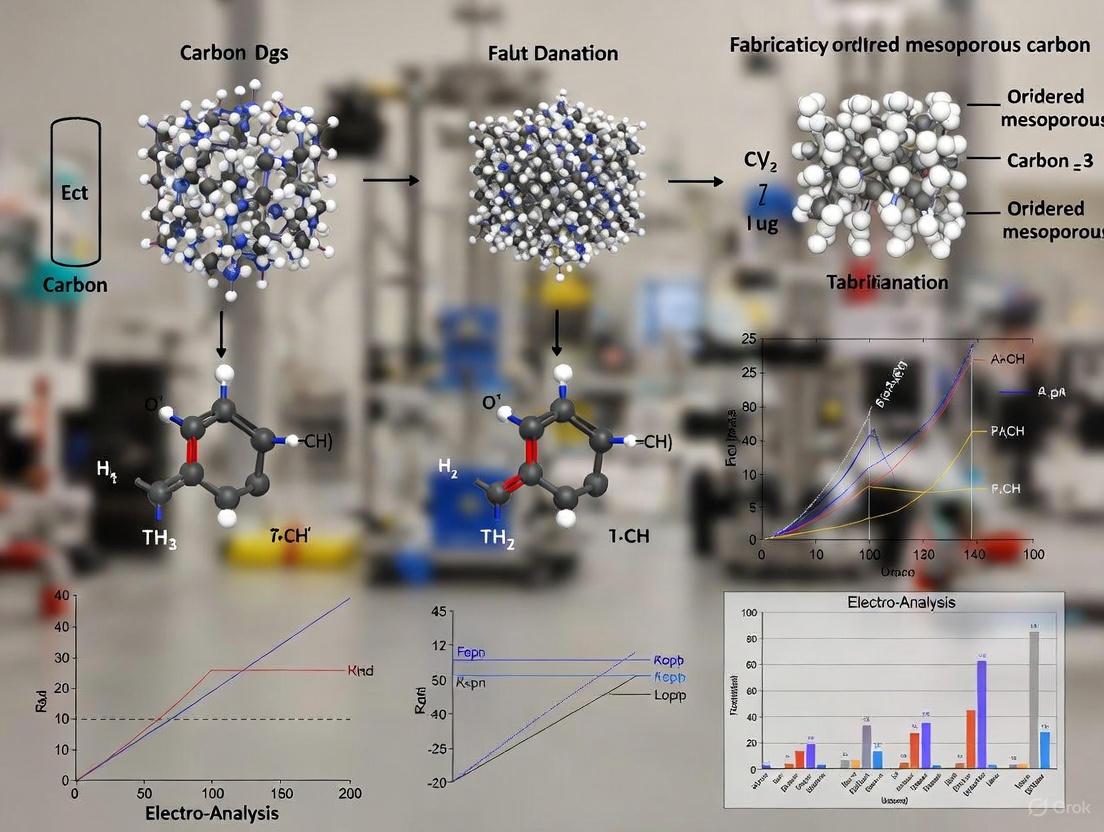
Ordered mesoporous carbon materials, particularly those with three-dimensional (3D) cubic structures, have emerged as a promising platform for advanced biomedical applications, most notably in drug delivery systems (DDS) [22] [24]. Their appeal in biomedical environments stems from a unique set of physicochemical properties—high specific surface area, large pore volume, tunable pore size distribution, and interconnected porous networks—which directly govern their interactions with biological entities and therapeutic molecules [3]. The fabrication of 3D ordered mesoporous carbons, such as CMK-8 and CMK-9, via nanocasting techniques using mesoporous silica templates, allows for precise structural control that can be tailored to specific biomedical requirements [22] [3]. Understanding the relationship between their nanoscale architecture (structure) and their performance as drug carriers (property) is fundamental to designing more effective and controlled therapeutic interventions. This application note details these critical structure-property relationships, provides validated protocols for their evaluation, and visualizes the key workflows and dependencies essential for researchers in the field.
Structural Characteristics and Comparative Properties
The structural parameters of 3D ordered mesoporous carbons are primary determinants of their drug loading capacity and release kinetics. The following table summarizes the key structural characteristics and resulting properties for two prominent 3D mesoporous carbons, CMK-8 and CMK-9, which are crucial for their performance in biomedical environments.
Table 1: Structural Characteristics and Drug Delivery Properties of 3D Ordered Mesoporous Carbons
| Material | Synthesis Precursor | Surface Area (m²/g) | Pore Characteristics | Cephalexin Adsorption Capacity (mg/g) | CFX Release at pH 1.2 (14 h) | Proposed Drug Release Mechanism |
|---|---|---|---|---|---|---|
| CMK-8 | Sucrose [22] | ~1000 [22] | Interconnected branched rods, intertubular mesopores [22] | 339 [22] | Information Missing | Fickian diffusion [22] |
| CMK-9 | Furfuryl Alcohol [22] | 1130 [22] | Dual mesoporosity (intra- and inter-tubular mesopores) [22] | 354 [22] | 89% [22] | Fickian diffusion [22] |
The data illustrates a direct structure-property relationship: CMK-9's superior surface area and more complex dual mesoporosity contribute to its higher drug adsorption capacity and enhanced controlled release performance compared to CMK-8 [22].
Experimental Protocols
Protocol: Synthesis of CMK-8 via Hard-Templating
This protocol outlines the steps for synthesizing CMK-8 using KIT-6 mesoporous silica as a hard template [22].
- Template Preparation: Begin with 1 gram of synthesized KIT-6 silica template [22].
- Precursor Solution Preparation: In a separate container, prepare a solution containing 1.86 g of sucrose (carbon precursor), 0.21 g of sulfuric acid (H₂SO₄, catalyst), and 7.3 g of deionized water [22].
- Template Infiltration: Slowly disperse the KIT-6 powder into the precursor solution under continuous stirring at room temperature. Continue stirring for 3 hours to ensure thorough infiltration of the template pores [22].
- Polymerization & Carbonization: Transfer the mixture to an oven and heat to 100°C for 6 hours, followed by 160°C for an additional 6 hours. This step polymerizes and partially carbonizes the sucrose within the template. For complete carbonization, subsequently heat the composite to 900°C under an inert atmosphere (e.g., nitrogen or argon) and hold for 2 hours [22].
- Template Removal: After cooling, immerse the silica-carbon composite in a hydrofluoric acid (HF) or sodium hydroxide (NaOH) solution to selectively etch away the KIT-6 silica template [22].
- Product Recovery: Filter the resulting black powder (CMK-8), wash thoroughly with deionized water and ethanol, and dry to obtain the final product [22].
Protocol: Surface Functionalization with APTES
Functionalization with 3-aminopropyltriethoxysilane (APTES) introduces amine groups, enhancing aqueous dispersion and providing interaction sites for controlled drug release [22].
- Material Pre-drying: Activate the surface of the synthesized mesoporous carbon (e.g., CMK-8 or CMK-9) by heating under vacuum to remove adsorbed water.
- Reaction Mixture Preparation: Disperse the dried carbon material in a suitable anhydrous solvent (e.g., toluene). Add a calculated volume of APTES (typically 1-10% v/v) to the suspension [22].
- Grafting Reaction: Reflux the mixture under an inert atmosphere (e.g., nitrogen) for a defined period (e.g., 24 hours) to allow the silane groups to covalently graft onto the carbon surface [22].
- Product Purification: After cooling, filter the functionalized material and wash repeatedly with the same solvent and ethanol to remove any physisorbed APTES.
- Drying: Dry the final product (denoted as, for example, C8-NH₂ or C9-NH₂) under vacuum [22].
Protocol: Drug Loading and In Vitro Release Kinetics
This protocol describes a standard method for evaluating the drug delivery potential of mesoporous carbons using cephalexin (CFX) as a model antibiotic [22].
Drug Loading via Adsorption:
- Prepare a concentrated solution of cephalexin in a suitable solvent.
- Disperse a known mass of the mesoporous carbon (pristine or functionalized) in the drug solution.
- Stir the suspension for 24 hours at room temperature to reach adsorption equilibrium.
- Separate the drug-loaded carbon via centrifugation or filtration.
- Wash the solid gently to remove surface-adsorbed drug and dry to obtain the final drug-loaded carrier. The drug loading capacity can be determined by measuring the concentration difference in the supernatant before and after adsorption using UV-Vis spectroscopy [22].
In Vitro Release Study:
- Place a known quantity of the drug-loaded carbon into a dialysis membrane or directly suspend it in the release medium.
- Use standardized biorelevant media, such as simulated gastric fluid (pH 1.2) and simulated intestinal fluid (pH 6.8), maintained at 37°C under constant agitation [22].
- At predetermined time intervals, withdraw a sample of the release medium and replace it with an equal volume of fresh medium to maintain sink conditions.
- Analyze the drug concentration in the withdrawn samples using HPLC or UV-Vis spectroscopy [22].
- Model the release data using kinetic models (e.g., Weibull model) to understand the release mechanism [22].
Visualization of Workflows and Relationships
The following diagrams, generated with Graphviz, illustrate the logical workflow of material synthesis and application, and the core structure-property relationships that define their biomedical function.
Synthesis to Application Workflow
Structure-Property Relationship Map
The Scientist's Toolkit: Essential Research Reagents and Materials
The table below catalogs key reagents, materials, and their functions essential for research on 3D ordered mesoporous carbons in biomedical applications.
Table 2: Key Research Reagent Solutions and Materials
| Item | Function / Role | Application Note |
|---|---|---|
| KIT-6 Mesoporous Silica | Hard template for nanocasting; defines 3D pore structure [22]. | The pore size and symmetry of KIT-6 directly dictate the resulting carbon's architecture [22]. |
| Sucrose / Furfuryl Alcohol | Carbon precursor; fills template pores to form carbon framework [22]. | Choice of precursor influences carbon wall properties and final porosity (e.g., CMK-8 vs. CMK-9) [22]. |
| APTES (3-Aminopropyltriethoxysilane) | Surface functionalization agent; introduces amine groups for controlled release [22]. | Enhances hydrophilicity and enables pH-dependent electrostatic interactions with drug molecules [22]. |
| Cephalexin (CFX) | Model drug molecule; a β-lactam antibiotic with a short half-life [22]. | Ideal for demonstrating sustained release to overcome limitations of conventional administration [22]. |
| Simulated Biological Fluids (pH 1.2, 6.8) | In vitro release media; mimics physiological conditions for performance testing [22]. | Critical for evaluating the material's release profile and kinetics in a biorelevant environment [22]. |
Advanced Synthesis and Functionalization for Targeted Drug Delivery
The fabrication of three-dimensionally (3D) ordered mesoporous carbons represents a significant frontier in materials science, with particular importance for applications requiring high surface area and controlled pore architecture, such as drug delivery, adsorption, and electrocatalysis. Among these materials, CMK-8 and CMK-9 carbons, synthesized through the nanocasting of KIT-6 mesoporous silica, offer distinctive 3D cubic structures with interconnected pore networks that facilitate molecular transport and accessibility. This protocol details the synthesis, functionalization, and application of these materials, providing a standardized framework for researchers engaged in the development of advanced mesoporous systems. The methods outlined herein are contextualized within a broader thesis on fabricating 3D-ordered mesoporous carbons, emphasizing the critical relationship between synthetic parameters, final material properties, and functional performance in targeted applications such as controlled drug release.
Material Characteristics and Comparative Analysis
CMK-8 and CMK-9 are both 3D cubic ordered mesoporous carbons with Ia3d symmetry, replicated from a KIT-6 silica hard template. Their structural differences originate from the carbon precursor and synthesis conditions, leading to distinct textural properties that influence their application performance [22].
- CMK-8 is typically synthesized using sucrose as a carbon precursor, forming a 3D network of interconnected branched rods with intertubular mesopores [22].
- CMK-9 is synthesized from furfuryl alcohol, resulting in a dual mesoporous system consisting of both intratubular mesopores within the carbon nanotubes and intertubular mesopores created after silica removal [22].
The table below summarizes the key characteristics of these materials and their performance in drug delivery applications.
Table 1: Structural Properties and Drug Delivery Performance of CMK-8 and CMK-9
| Feature | CMK-8 | CMK-9 |
|---|---|---|
| Carbon Precursor | Sucrose [22] | Furfuryl alcohol [22] |
| Mesoporous Structure | 3D network of interconnected rods; intertubular mesopores [22] | Dual mesoporosity (intra- and inter-tubular mesopores) [22] |
| Specific Surface Area | >500 m²/g [25] | 1130 m²/g [22] |
| Pore Diameter | 3.2–6.6 nm [25] | Information not specified in search results |
| Cephalexin Adsorption Capacity | 339 mg/g [22] | 354 mg/g [22] |
| Cephalexin Release (pH 1.2, 14 h) | Information not specified in search results | 89% [22] |
| Primary Release Mechanism | Fickian diffusion [22] | Fickian diffusion [22] |
Experimental Protocols
Synthesis of KIT-6 Silica Hard Template
The synthesis of the KIT-6 silica template is a critical first step, as its quality directly determines the structural fidelity of the final carbon replica [4] [26].
- Reagents: Pluronic P123 (EO₂₀PO₇₀EO₂₀), tetraethyl orthosilicate (TEOS), concentrated hydrochloric acid (HCl, 35 wt%), n-butanol, deionized water [4] [26].
- Procedure:
- Dissolve 5.0 g of Pluronic P123 in a mixture of 180 g of deionized water and 9.9 g of concentrated HCl under vigorous stirring at 35 °C until a clear solution is obtained [4] [26].
- Add 5.0 g of n-butanol to the solution and continue stirring for 1 hour [4].
- Rapidly add 10.75 g of TEOS to the homogeneous solution and maintain stirring at 35 °C for 24 hours [4].
- Transfer the mixture into a sealed autoclave and age at 100 °C under static conditions for an additional 24 hours [4].
- Filter the resulting solid product and dry it overnight at 100 °C [4].
- Calcine the dried powder at 550 °C for 6 hours in air to remove the organic template, yielding the final KIT-6 silica with a cubic Ia3d structure [4] [26].
Synthesis of CMK-8 Carbon
This protocol describes the synthesis of CMK-8 using sucrose as the carbon precursor, following a two-step impregnation process [22] [4].
- Reagents: KIT-6 silica, sucrose, sulfuric acid (H₂SO₄), deionized water [22] [4].
- Procedure:
- Prepare an impregnation solution by dissolving 1.86 g of sucrose and 0.21 g of H₂SO₄ in 7.3 g of deionized water [22].
- Slowly disperse 1.0 g of KIT-6 silica into the solution and stir at room temperature for 3 hours to ensure thorough pore infiltration [22].
- Heat the mixture at 100 °C for 6 hours, followed by further heating at 160 °C for 6 hours [22] [4].
- Repeat the impregnation step with a second solution of identical composition (1.86 g sucrose, 0.21 g H₂SO₄, 7.3 g water) and repeat the heat treatment at 100 °C and 160 °C [22] [4].
- Carbonize the resulting dark brown composite in a tube furnace at 900 °C for 3 hours under an inert nitrogen atmosphere [22] [4].
- Remove the silica template by stirring the carbonized composite in a 5 wt% hydrofluoric (HF) acid solution or a 10 M sodium hydroxide (NaOH) solution at room temperature [22] [4].
- Filter, wash thoroughly with deionized water and ethanol, and dry to obtain the final CMK-8 carbon [22].
Synthesis of CMK-9 Carbon
CMK-9 is synthesized using furfuryl alcohol as the precursor, which requires an polymerization step inside the silica pores [22].
- Reagents: KIT-6 silica, furfuryl alcohol, trimethylbenzene (TMB), oxalic acid, acetone [22].
- Procedure:
- Impregnate 1.0 g of KIT-6 silica with a mixture of furfuryl alcohol and TMB (mass ratio 10:3) [22].
- Add an aqueous oxalic acid solution (0.5 M) to catalyze the polymerization of furfuryl alcohol within the pores of KIT-6 [22].
- Allow the mixture to stand for 24 hours to complete the polymerization process [22].
- Wash the resulting composite with acetone to remove any excess precursor and dry [22].
- Carbonize the powder at 900 °C for 3 hours under a nitrogen atmosphere [22].
- Remove the silica framework using HF or NaOH solution, as described for CMK-8 [22].
- Filter, wash, and dry the obtained CMK-9 carbon [22].
Surface Functionalization with APTES
Functionalization with 3-aminopropyltriethoxysilane (APTES) introduces amine groups, which enhance aqueous dispersion and influence drug release profiles [22].
- Reagents: CMK-8 or CMK-9 carbon, 3-aminopropyltriethoxysilane (APTES), anhydrous toluene [22].
- Procedure:
- Disperse 0.5 g of the mesoporous carbon in 50 mL of anhydrous toluene [22].
- Add 2.5 mL of APTES to the suspension and reflux under an inert atmosphere at 110 °C for 24 hours with continuous stirring [22].
- Filter the functionalized solid and wash sequentially with toluene, ethanol, and deionized water to remove any unreacted silane [22].
- Dry the final product (denoted as C8-N or C9-N) at 80 °C overnight [22].
Drug Loading and In Vitro Release Protocol
This protocol uses the antibiotic cephalexin (CFX) as a model drug to evaluate the materials' performance as nanocarriers [22].
- Reagents: Pristine or functionalized CMK-8/CMK-9, cephalexin (CFX), hydrochloric acid (HCl), potassium phosphate buffer [22].
- Drug Loading:
- In Vitro Release Study:
- Place the drug-loaded carbon into a dissolution apparatus containing a buffer medium (e.g., pH 1.2 for simulated gastric fluid or pH 6.8 for simulated intestinal fluid) at 37 °C under continuous agitation [22].
- Withdraw samples at predetermined time intervals and analyze the drug concentration using UV-Vis spectroscopy or HPLC [22].
- Cumulative release percentages are calculated to generate release profiles, which can be fitted to kinetic models like the Weibull model to understand the release mechanism [22].
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for KIT-6, CMK-8, and CMK-9 Synthesis
| Reagent | Function in Synthesis | Critical Notes |
|---|---|---|
| Pluronic P123 | Structure-directing agent (soft template) for KIT-6 silica. Forms micelles around which silica condenses [4] [17]. | The PEO-PPO-PEO block copolymer is essential for creating the cubic Ia3d mesostructure [17]. |
| Tetraethyl Orthosilicate (TEOS) | Silica precursor for the KIT-6 hard template [4] [26]. | Hydrolyzes and condenses on the surface of P123 micelles to form the inorganic framework. |
| n-Butanol | Cosurfactant (swelling agent) in KIT-6 synthesis. Promotes the formation of the 3D cubic Ia3d structure over 2D hexagonal [4] [17]. | Concentration is critical for achieving the desired pore symmetry and connectivity. |
| Sucrose | Carbon precursor for CMK-8. Fills the pores of KIT-6 and converts to carbon during pyrolysis [22] [4]. | Low-cost and common precursor; requires H₂SO₄ as a dehydration catalyst. |
| Furfuryl Alcohol | Carbon precursor for CMK-9. Polymerizes inside the KIT-6 pores before carbonization [22]. | Leads to the formation of the distinct dual mesoporosity in CMK-9. |
| APTES | Surface functionalization agent. Introduces terminal amine (-NH₂) groups to the carbon surface [22]. | Enhances hydrophilicity and enables controlled drug release via electrostatic interactions. |
| Boric Acid (H₃BO₃) | Pore expanding agent. Can be added to the carbon precursor to systematically increase the pore size of the final carbon replica [4]. | Allows for pore size tuning in the range of ~3.9 to 9.4 nm [4]. |
Workflow and Structural Visualization
The synthesis of CMK-8 and CMK-9 via the hard-template method involves a multi-step sequence of template preparation, precursor infiltration, and template removal, leading to distinct structural outcomes.
Synthesis Pathway for CMK-8 and CMK-9
The molecular interactions and drug release mechanisms within the mesoporous carbon nanocarriers are governed by diffusion and surface chemistry.
Drug Loading and Release Mechanism
Application in Controlled Drug Release
The 3D porous architecture of CMK-8 and CMK-9 makes them exceptional candidates for drug delivery systems (DDS). Their performance is demonstrated using cephalexin (CFX), a short-half-life antibiotic, as a model drug [22].
- Superior Loading and Release: CMK-9 exhibits a higher CFX adsorption capacity (354 mg/g) than CMK-8 (339 mg/g), attributed to its larger surface area and dual mesoporosity, which provide more adsorption sites and facilitate drug diffusion [22]. Under simulated gastric conditions (pH 1.2), the APTES-functionalized CMK-9 (C9-N) achieves 89% CFX release within 14 hours, significantly outperforming pure crystalline CFX, which releases its content too rapidly [22].
- Release Kinetics and Mechanism: The release profiles for both carbons fit the Weibull model, indicating a Fickian diffusion-controlled release mechanism [22]. This means drug release is primarily governed by concentration gradients and the intricate pore pathways of the carbon matrix. Surface functionalization with APTES further modulates the release by introducing electrostatic interactions between the amine groups on the carbon and the drug molecules [22].
The hard-template synthesis using KIT-6 silica provides a robust and versatile pathway for fabricating 3D ordered mesoporous carbons with well-defined pore architectures. As detailed in these Application Notes, CMK-8 and CMK-9 represent two structurally distinct outcomes from this pathway, with CMK-9's dual mesoporosity rendering it particularly effective for controlled drug delivery applications. The protocols for synthesis, functionalization, and drug loading/release provide a comprehensive toolkit for researchers. Future work in this domain, as guided by this thesis, should focus on further pore engineering, scaling production, and exploring synergistic multi-functionalization to unlock the full potential of these materials in biomedicine and beyond.
The fabrication of three-dimensionally ordered mesoporous carbon (3DOMC) materials represents a cornerstone of modern materials science, enabling advanced applications in catalysis, energy storage, and drug delivery. Among the various synthesis routes, soft-template methods and Direct Ink Writing (DIW) have emerged as powerful and versatile strategies. Soft-templating utilizes molecular self-assembly of block copolymers to create ordered porous networks, offering a streamlined, single-step synthesis pathway [27] [28]. In parallel, DIW, an additive manufacturing technique, provides unparalleled spatial control for structuring carbon-based composites into complex 3D architectures [29] [30]. This Application Note details standardized protocols for these methods, providing researchers with the foundational tools for fabricating next-generation 3DOMC materials.
Soft-Template Synthesis of Ordered Mesoporous Carbon
The soft-template approach relies on the self-assembly of amphiphilic block copolymers to structure a carbon precursor. The method involves co-dissolving the template and precursor, followed by evaporation-induced self-assembly (EISA), thermal polymerization, and finally, carbonization to yield the porous carbon structure [27] [28].
Experimental Protocol: Gram-Scale Synthesis of N-Doped Hollow Nanoporous Carbon Spheres
The following protocol, adapted from a gram-scale production method, yields nitrogen-doped hollow nanoporous carbon spheres (N-HNCS) using a silica-assisted soft-template approach [31].
- 1. Solution Preparation: Dissolve 5.558 g of Cetyltrimethylammonium bromide (CTAB) in a mixture of 180 mL deionized water (DIW) and 100 mL ethanol (EtOH). Add 1 mL of ammonia aqueous solution (NH₄OH) under stirring at 70°C.
- 2. Resorcinol Addition: Introduce a pre-mixed solution of 1.1 g resorcinol in 20 mL DIW to the reaction vessel. Stir for 30 minutes.
- 3. Silica Incorporation and Polymerization: Add 6 mL tetraethylorthosilicate (TEOS) and 1.48 mL formaldehyde (37%) to the solution. Subsequently, add 0.6 g melamine and 1.1 mL formaldehyde to incorporate nitrogen. Stir the reaction mixture continuously for 24 hours at 70°C.
- 4. Product Recovery: Filter the resulting dark orange solid (MRF resin-silica spheres). Wash thoroughly with DIW and EtOH, then air-dry the product overnight at 60°C.
- 5. Pyrolysis (Carbonization): Place the dried powder in a tube furnace under an inert argon atmosphere.
- Heat to 350°C at a rate of 3°C/min and hold for 3 hours.
- Subsequently, raise the temperature to 800°C at 2.5°C/min and maintain for 2 hours.
- Allow the sample to cool to room temperature.
- 6. Template Removal: Disperse the black carbon/silica composite in a hydrofluoric acid (HF) solution (1:4.5:4.5 volume ratio of HF:DIW:EtOH) to etch the silica. Stir for 1.5 hours, then filter, wash with copious amounts of DIW and EtOH, and dry at 60°C to obtain the final N-HNCS product (yield: ~1.1 g) [31].
Synthesis Workflow and Key Parameters
The diagram below illustrates the procedural workflow and critical synthesis parameters for the soft-templating method.
Material Properties and Optimization
The properties of the resulting mesoporous carbon are highly tunable by adjusting synthesis conditions. The table below summarizes key characteristics and the effects of varying common parameters, as demonstrated in the literature [27] [31] [7].
Table 1: Properties and Optimization of Soft-Templated Mesoporous Carbons
| Material Property | Typical Performance / Range | Influence of Synthesis Parameters |
|---|---|---|
| Specific Surface Area | Up to 1250 m²/g [31] | Increased by higher carbonization temperature and efficient template removal. |
| Pore Volume | ~1.2 cm³/g [31] | Determined by the template-to-precursor ratio and the structure of the self-assembled mesophase. |
| Pore Size | 2-50 nm (mesopore range) [3] | Directly controlled by the molecular weight of the hydrophobic block of the polymer template (e.g., Pluronic F127, P123) [27] [28]. |
| Nitrogen Doping | Achieved via melamine precursor [31] | Enhances electrochemical activity and surface polarity. Doping level controlled by precursor amount. |
| Conductivity | Highly dependent on carbonization temperature [28] | Higher carbonization temperatures (e.g., 800-900°C) improve electrical conductivity and mechanical stability [27]. |
Direct Ink Writing of Carbon Composites
DIW enables the fabrication of complex 3D structures by extruding a shear-thinning carbon composite ink in a layer-by-layer fashion. The success of this method hinges on formulating an ink with optimal rheology [29] [30].
Experimental Protocol: DIW of Carbon Fiber Composite Inks
This protocol outlines the preparation and printing of a carbon fiber-reinforced thermoset polymer ink [29] [30].
- 1. Ink Formulation: Prepare a composite ink by uniformly dispersing chopped carbon fibers (CFs) within a thermoset polymer matrix (e.g., epoxy). The ink must contain rheology modifiers like nanoclay or fumed silica (typically 1-5 wt%) to induce shear-thinning behavior and a yield stress, ensuring the printed filament retains its shape after deposition [30].
- 2. Rheology Check: Confirm the ink exhibits viscous flow under shear (extrusion) but rapidly solidifies at rest. This is typically verified using a rheometer to measure storage (G') and loss (G") moduli [29].
- 3. Printer Setup: Load the ink into a syringe barrel. Attach a nozzle with a diameter selected based on the desired print resolution (e.g., 200-840 µm). Ensure the printing stage is level and the substrate is clean and secure.
- 4. Printing Parameters Optimization: Calibrate the following key parameters for print fidelity:
- Extrusion Pressure: Adjust to achieve a consistent filament flow without dripping or under-extrusion.
- Print Speed: Synchronize with material extrusion rate.
- Layer Height: Typically 50-90% of the nozzle diameter [30].
- Step-Over Distance: The distance between adjacent toolpaths, affecting inter-filament bonding and surface finish.
- 5. Post-Processing: After printing, cure the structure as required by the polymer matrix (e.g., UV cure for photoresins or thermal cure for epoxies) to achieve its final mechanical properties [29].
DIW Process and Parameter Interplay
The following diagram visualizes the DIW process workflow and highlights the critical interrelationships between ink properties, printer parameters, and final print quality.
DIW Ink Compositions and Print Parameters
The table below summarizes common carbon materials used in DIW and the typical print parameters that influence the structural outcome, crucial for achieving high-fidelity 3D structures [29] [30].
Table 2: DIW Carbon Inks and Optimized Printing Parameters
| Category | Component / Parameter | Example / Optimal Range | Function / Impact |
|---|---|---|---|
| Carbon Fillers | Carbon Nanotubes (CNTs) | 0.5-5 wt% | Enhances electrical conductivity and mechanical strength. High aspect ratio requires good dispersion. |
| Graphene Oxide (GO) | 1-10 mg/mL | Provides a 2D sheet structure for mechanical reinforcement. Can be reduced post-print (rGO) for conductivity. | |
| Carbon Fibers (CFs) | 10-30 wt% (chopped) | Primary reinforcement for high strength and stiffness. Affects viscosity and fiber alignment during extrusion [30]. | |
| Polymer Matrix | Epoxy, PLA, Thermoset Resins | Continuous phase | Provides bulk form, protects reinforcements, and enables load transfer. |
| Additives | Nanoclay, Fumed Silica | 1-5 wt% | Acts as a rheology modifier to impart shear-thinning behavior and prevent slumping [30]. |
| Print Parameters | Nozzle Diameter | 200 - 840 µm | Determines printing resolution and minimum feature size. |
| Layer Height | 50 - 90% of nozzle diameter | Affects inter-layer adhesion and Z-axis resolution [30]. | |
| Step-Over Distance | 80 - 100% of nozzle width | Influences wall density and surface roughness. |
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for 3DOMC Fabrication
| Reagent / Material | Function | Specific Examples |
|---|---|---|
| Block Copolymer Templates | Structure-directing agent; forms the mesoporous framework via self-assembly. | Pluronic F127 (PEO-PPO-PEO), CTAB [27] [31] [28]. |
| Carbon Precursors | Source of carbon; undergoes polymerization and pyrolysis to form the carbon matrix. | Phenolic resol, resorcinol-formaldehyde (RF) resin, sucrose [27] [28] [7]. |
| Dopant Precursors | Introduces heteroatoms into the carbon lattice to modify electronic and chemical properties. | Melamine (for N-doping) [31]. |
| Rheology Modifiers | Imparts shear-thinning behavior to DIW inks for shape retention. | Nanoclay, fumed silica [30]. |
| Solvents | Medium for dissolution and self-assembly of precursors and templates. | Ethanol, deionized water, methanol [31] [28]. |
| Etchants | Removes template material (soft or hard) to liberate the porous carbon structure. | Hydrofluoric Acid (HF) for silica [31] [7]. |
The integration of three-dimensionally ordered mesoporous carbons (3DOMCs) into structurally hierarchical scaffolds represents a frontier in materials science, offering unparalleled control over material properties at multiple length scales. This protocol details the emerging methodology of one-pot 3D printing for fabricating such scaffolds, which combine the high surface area and tailored pore connectivity of 3DOMCs with the macroscopic shape fidelity and mechanical integrity afforded by additive manufacturing. Developed for researchers and drug development professionals, this document provides a complete framework—from material synthesis and ink formulation to computational design and post-processing—enabling the creation of advanced scaffolds for applications in catalysis, energy storage, and controlled drug delivery. The synthesized architectures demonstrate significant enhancements in mass transfer, active site accessibility, and functional performance, as quantified in the accompanying data.
Quantitative Performance Data of Hierarchical Porous Scaffolds
The tables below summarize key performance metrics for various hierarchical porous materials and 3D-printed scaffolds, highlighting their enhanced properties.
Table 1: Textural and Electrochemical Properties of Hierarchical Porous Carbons
| Material | BET Surface Area (m²/g) | Pore Volume (cm³/g) | Primary Pore Size | Electrochemical Performance | Application |
|---|---|---|---|---|---|
| HOM-AMUW Carbon [32] | 451 | N/D | Macropores: ~250 nm; Walls: 4-5 nm (mesoporous) | Discharge capacity: 12,686 mAh/g at 2000 mA/g | Li-O₂ Battery Cathode |
| Hierarchical Biobased Carbon [33] | 558 | N/D | Macro/Meso/Micro | Specific capacitance: 182 F/g | Supercapacitor |
| ACRKB (Biomass-Derived) [34] | 568 | N/D | Hierarchical | Li–Se capacity: 470.76 mAh/g after 400 cycles | Li–Se / Na–Se Batteries |
| CMK-9 [22] | 1130 | N/D | Dual Mesoporosity (3D Cubic) | Cephalexin adsorption: 354 mg/g; 89% release in 14h | Drug Delivery |
Table 2: Performance Metrics of 3D-Printed Functional Scaffolds
| Printed Material | Mechanical Strength | Key Functional Performance | Reusability/Cycling Stability | Application |
|---|---|---|---|---|
| TS-1 Monolith Catalyst [35] | Crush strength: 135.11 N/cm | Ethylene Glycol yield: 82.7%; H₂O₂ utilization: 85.3% | High performance over 5 cycles | One-pot synthesis |
| PL-uW Cryo-printed Scaffold [36] | N/D | Promoted vascularized bone regeneration, M1-to-M2 macrophage polarization | N/A | Bone Tissue Engineering |
| ZnO/OMCS [7] | N/D | Microwave absorption: -39.3 dB at 10.4 GHz; Bandwidth: 9.1 GHz | N/A | Electromagnetic Absorption |
Experimental Protocols
Protocol 1: One-Pot Fabrication of 3D-Printed TS-1 Monolithic Catalysts
This protocol describes the direct ink writing (DIW) of a structured SiO₂-based TS-1 catalyst with hierarchical porosity for catalytic applications like the one-pot synthesis of ethylene glycol [35].
Materials and Reagent Solutions
- Catalytic Active Phase: Titanium silicalite-1 (TS-1) powder (median particle size ~48.8 µm).
- Ink Matrix: Spherical silica powder (D50 = 2 µm) acts as the binder and matrix component.
- Plasticizing Binder: Sesbania powder (D50 = 139.8 µm) to enhance printability and green strength.
- Rheology Modifier: Polyethylene glycol (PEG6000) to impart suitable thixotropic behavior to the ink.
- Solvent: Deionized water.
- Equipment: Direct Ink Writing (DIW) 3D printer, mixer, sintering furnace.
Step-by-Step Procedure
- Ink Formulation: Thoroughly mix 40 g of TS-1 powder, 10 g of spherical silica powder, 1.5 g of sesbania powder, and 1.5 g of PEG6000. Gradually add 16 ml of deionized water while mixing to form a homogeneous, high-viscosity paste with excellent thixotropy [35].
- Biomimetic Design & CFD-Guided Optimization: Design the printing path to create a biomimetic spiral porous structure inspired by the nautilus shell. Use Computational Fluid Dynamics (CFD) to simulate the flow field and pressure drop within the target reactor, optimizing the monolith's channel offset angles and dimensions (axial and radial) for maximum catalytic efficiency [35].
- 3D Printing (Direct Ink Writing): Load the prepared ink into the DIW printer's syringe. Utilize a nozzle diameter of 1.0 mm to extrude the ink layer-by-layer according to the digital design. Maintain a constant printing speed and pressure to ensure filament uniformity and structural integrity [35].
- Post-Processing and Sintering: Carefully dry the printed "green" body at room temperature. Subsequently, sinter the monolith in a furnace at 550°C. This critical step removes organic binders and consolidates the silica matrix, finalizing the hierarchical porous structure and imparting mechanical strength [35].
- Quality Control: The sintered scaffold should exhibit a crush strength of >135 N/cm.
Protocol 2: Synthesis of 3D Cubic Ordered Mesoporous Carbon (CMK-8/9)
This protocol outlines the hard-template synthesis of 3D cubic ordered mesoporous carbons (OMCs) like CMK-8 and CMK-9, which are ideal nanocarriers for drug delivery given their high surface area and tunable pore sizes [22] [4].
Materials and Reagent Solutions
- Hard Template: KIT-6 mesoporous silica with cubic Ia3d symmetry.
- Carbon Precursors: Sucrose (for CMK-8) or Furfuryl alcohol (for CMK-9).
- Catalyst: Sulfuric acid (H₂SO₄).
- Pore Expanding Agent (Optional): Boric acid, for tailoring the final pore size [4].
- Etching Agent: Hydrofluoric acid (HF) solution (5 wt%) or sodium hydroxide (NaOH) solution.
Step-by-Step Procedure
- Template Impregnation:
- For CMK-8: Dissolve 1.25 g of sucrose, 0.142 g of H₂SO₄, and (optionally) the desired amount of boric acid in 5 g of water. Add 1 g of KIT-6 silica to this solution and mix thoroughly. Heat the mixture at 100°C for 6 hours, followed by 160°C for another 6 hours [4].
- For CMK-9: Impregnate the KIT-6 silica with furfuryl alcohol, which undergoes polymerization inside the mesopores [22].
- Carbonization: Place the impregnated silica/composite in a tube furnace. Carbonize under an inert nitrogen atmosphere with a temperature program: ramp to 350°C to remove surfactants, then increase to 900°C and hold for 2-3 hours to convert the precursor to glassy carbon [22] [4].
- Template Removal: After carbonization, remove the silica template by washing the composite with a 5 wt% HF solution twice, or with a NaOH solution (e.g., 1M or 2M). Recover the resulting ordered mesoporous carbon powder by filtration, followed by washing with ethanol and water, and final drying [22] [4].
- Expected Outcome: CMK-9 typically exhibits dual mesoporosity and a higher surface area (~1130 m²/g) than CMK-8, leading to superior drug loading capacity (e.g., 354 mg/g for cephalexin) [22].
Protocol 3: Fabrication of Hierarchical Carbon-Carbon Composite Monoliths
This method uses cellulosic fiber fabric as a structuring agent to create mechanically robust, self-standing hierarchical carbon monoliths without complex solvent exchange [33].
Materials and Reagent Solutions
- Macroporous Scaffold: Woven cellulosic fiber fabric (e.g., raw cotton canvas).
- Mesoporous Resin Precursors: Resorcinol and Formaldehyde.
- Catalyst & Stabilizer: Sodium carbonate (Na₂CO₃) and Cetyltrimethylammonium bromide (CTAB).
- Equipment: Oven, tube furnace, mixing frames.
Step-by-Step Procedure
- Resin Synthesis and Fabric Impregnation: Prepare a viscous prepolymer solution with a Resorcinol/Formaldehyde/Catalyst molar ratio of 1:2.9:0.005. Impregnate the thoroughly washed and stretched cellulosic fabric with this solution until saturated [33].
- Curing and Drying: Cure the impregnated fabric at 70°C for 24 hours in a water-saturated atmosphere to cross-link the resin. Subsequently, dry the composite at ambient pressure. The cellulosic fibers act as a stabilizer, preventing the collapse of the resin's mesoporosity during air drying [33].
- Carbonization: Carbonize the cured composite in an inert atmosphere at 900°C. This process converts the resorcinol-formaldehyde resin and the cellulosic fabric into an interconnected carbon network, resulting in a monolithic carbon-carbon composite [33].
- Key Advantage: This method simultaneously creates micro/mesopores (from the resin) and macropores (from the fabric weave and fiber structure), yielding a hierarchical pore system with a BET surface area of ~558 m²/g and improved mechanical properties due to the carbonized fiber reinforcement [33].
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagent Solutions for Fabricating Hierarchical Porous Scaffolds
| Reagent / Material | Function / Role | Example Application / Note |
|---|---|---|
| KIT-6 Mesoporous Silica | A hard template with 3D bicontinuous cubic (Ia3d) structure for replicating ordered mesopores. | Synthesis of CMK-8 and CMK-9 carbons [22] [4]. |
| Pluronic P123 Triblock Copolymer | A structure-directing agent (SDA) used in the synthesis of mesoporous silica templates like KIT-6 and SBA-15. | Forms the mesostructure via evaporation-induced self-assembly (EISA) [4]. |
| Resorcinol-Formaldehyde (RF) Resin | A versatile carbon precursor for producing synthetic mesoporous carbons. | Used in sol-gel processes to create carbon gels and composites [33]. |
| Cetyltrimethylammonium Bromide (CTAB) | A cationic surfactant used as a SDA or pore stabilizer. | Stabilizes mesopores in RF gels during air drying, preventing collapse [33]. |
| Boric Acid (H₃BO₃) | A pore expanding agent for tailoring the pore size of ordered mesoporous carbons. | Added to the carbon precursor; pore size increases with boric acid content [4]. |
Workflow and Conceptual Diagrams
The following diagrams illustrate the core fabrication workflows and structural relationships central to this field.
Within the scope of advanced material fabrication, the development of three-dimensionally ordered mesoporous carbon (3DOMC) represents a significant breakthrough. These materials are characterized by a highly interconnected mesopore system, large specific surface area (>1000 m²/g), high mesopore volume (>3 cm³/g), and stable carbon skeleton, making them exceptionally suitable for applications in adsorption, catalysis, and biomedicine [37] [38] [39]. However, the inherent hydrophobicity and neutral surface of pristine mesoporous carbon significantly limit its dispersion stability in biological and aqueous media, thereby restricting its effectiveness in controlled release applications [37].
Surface functionalization via (3-aminopropyl)triethoxysilane (APTES) grafting has emerged as a powerful strategy to overcome these limitations. This process transforms the neutral and hydrophobic carbon surface into a more hydrophilic one and introduces reactive amine groups (-NH₂) that serve as anchoring points for further bioconjugation [37] [40]. The covalent attachment of aminosilane molecules to the carbon framework not only enhances dispersion stability in polar solvents but also enables the construction of pH-responsive drug delivery systems through the formation of acid-labile imine bonds [40]. This protocol details the methodologies for the synthesis, functionalization, and application of APTES-grafted 3DOMC, providing researchers with a comprehensive framework for developing advanced drug delivery platforms.
Synthesis of Three-Dimensionally Ordered Mesoporous Carbon (3DOMC)
The fabrication of 3DOMC with a face-centered cubic close-packed structure typically employs a hard-template method using mesoporous silica as a solid matrix [37] [38]. The following protocol outlines a representative synthesis procedure.
Experimental Protocol: Hard-Template Synthesis of 3DOMC
Principle: This method utilizes ordered mesoporous silica (KIT-6) as a nanoreactor template. The mesopores are infiltrated with a carbon precursor, which is subsequently carbonized. The silica framework is then etched away, yielding an inverse carbon replica with a highly ordered 3D mesoporous structure [37] [38].
Materials:
- Mesoporous silica template (KIT-6)
- Carbon precursor: Sucrose
- Catalytic solution: Sulfuric acid (H₂SO₄)
- Ethanol
- Hydrofluoric acid (HF) or Sodium hydroxide (NaOH) for silica removal
Procedure:
- Impregnation: Dissolve 1.25 g of sucrose and 0.14 g of H₂SO₄ in 5 g of deionized water. Add 1.0 g of the mesoporous silica template (KIT-6) to this solution.
- Polymerization: Sonicate the mixture for 30 minutes to ensure thorough pore infiltration, then heat at 100 °C for 6 hours, followed by a further 6 hours at 160 °C.
- Carbonization: Place the resulting dark brown composite in a tubular furnace and heat under an inert atmosphere (Ar or N₂) to 800-900 °C at a heating rate of 5 °C min⁻¹. Maintain this temperature for 2-3 hours to achieve complete carbonization.
- Template Removal: After cooling, remove the silica framework by etching with a 5% HF solution for 3 days or with a hot, concentrated NaOH solution.
- Washing and Drying: Wash the resulting 3DOMC material thoroughly with ethanol and water, and dry overnight at 80-100 °C [37] [38].
The successful formation of the 3DOMC structure should be confirmed by scanning electron microscopy (SEM) and nitrogen physisorption analysis, which will reveal the highly ordered porous array and type-IV isotherm characteristic of mesoporous materials.
Research Reagent Solutions
Table 1: Essential Reagents for 3DOMC Synthesis and APTES Functionalization.
| Reagent | Function/Role in the Protocol |
|---|---|
| Mesoporous Silica (KIT-6) | Hard template to provide the inverse 3D ordered structure for carbon replication. |
| Sucrose | Carbon source that undergoes pyrolysis to form the carbon framework within the template. |
| Sulfuric Acid (H₂SO₄) | Catalyst for the polymerization and carbonization of the sucrose precursor. |
| Hydrofluoric Acid (HF) | Etching agent for the selective removal of the silica template after carbonization. |
| (3-Aminopropyl)triethoxysilane (APTES) | Aminosilane coupling agent that grafts amine-functional groups onto the oxidized carbon surface. |
| Hydrogen Peroxide (H₂O₂) or Nitric Acid (HNO₃) | Strong oxidizing agents used to generate surface hydroxyl groups on carbon for subsequent silane grafting. |
| Toluene or Methanol | Anhydrous solvents used as the reaction medium for the APTES grafting process. |
| Ninhydrin | Colorimetric assay reagent for the quantification of surface amine groups post-functionalization [40]. |
Surface Functionalization: APTES Grafting Protocol
The grafting of APTES onto the 3DOMC surface is a critical step that introduces amine functionalities, drastically altering the material's surface properties and enabling further conjugation.
Experimental Protocol: Surface Oxidation and APTES Grafting
Principle: The pristine carbon surface is first oxidized to generate hydroxyl and carboxyl groups, which serve as anchoring sites for silane molecules. APTES then undergoes hydrolysis and condensation with these surface groups, forming stable covalent Si-O-C bonds [37] [40].
Materials:
- Synthesized 3DOMC
- Oxidizing agent: Concentrated HNO₃ or H₂SO₄/H₂O₂ mixture
- (3-Aminopropyl)triethoxysilane (APTES)
- Anhydrous toluene
- Ethanol
Procedure:
- Surface Oxidation: Add 1 g of 3DOMC to 20 mL of concentrated nitric acid. Reflux the mixture at 120 °C for 2 hours to introduce surface oxygenated groups. Cool to room temperature.
- Washing: Dilute the mixture and isolate the oxidized carbon via centrifugation. Wash repeatedly with deionized water until the supernatant reaches a neutral pH. Dry the oxidized 3DOMC (now 3DOMC-ox) at 100 °C overnight.
- APTES Grafting: Disperse 0.5 g of 3DOMC-ox in 50 mL of anhydrous toluene by sonication for 30 minutes.
- Reaction: Add a specific volume of APTES (e.g., 1.2 mL to 6 mL, depending on the desired loading) to the dispersion. Carry out the functionalization reaction at 40 °C for 8 hours under continuous stirring [37].
- Purification: Recover the functionalized material (3DOMC-APTES) by centrifugation and wash extensively with ethanol to remove any physisorbed silane molecules.
- Drying: Dry the final product under vacuum at 60 °C for 12 hours.
Confirmation of Functionalization:
- FT-IR Spectroscopy: Look for the appearance of bands in the range of 960–1150 cm⁻¹, corresponding to Si-O-C stretching vibrations, and bands at ~2930 cm⁻¹ and ~2860 cm⁻¹ for the C-H stretching of the propyl chain of APTES [37].
- Elemental Analysis: An increase in nitrogen content confirms the successful incorporation of the aminopropyl group [37].
- Ninhydrin Assay: This colorimetric test can be used to quantify the surface amine density on the 3DOMC-APTES material [40].
- Thermogravimetric Analysis (TGA): The functionalized carbon will show distinct weight loss steps between 60–650 °C, corresponding to the degradation of the grafted aminosilane organic layer [37].
Visualization of the Functionalization and Drug Loading Workflow
The following diagram illustrates the sequential process from material synthesis to drug loading and release.
Workflow for 3DOMC-based Drug Delivery System
Application Notes: Enhanced Dispersion and Controlled Drug Release
The functionalization of 3DOMC with APTES profoundly impacts its performance in dispersion stability and controlled release applications, as quantified below.
Dispersion Stability in Various Solvents
The introduction of amine groups significantly alters the surface properties of 3DOMC. In polar solvents like water, the aminosilane-grafted carbons demonstrate superior stability compared to the unmodified material. The stability can be assessed using multiple light scattering techniques [37].
Table 2: Dispersion Stability of APTES-Grafted Mesoporous Carbons [37].
| Material | Dispersion Stability in Water | Dispersion Stability in Toluene | Dispersion Stability in Tween 80 Surfactant |
|---|---|---|---|
| Pristine 3DOMC | Low | Moderate | Low |
| 3DOMC-APTES (Low Load) | High | High | High |
| 3DOMC-APTES (High Load) | High | Moderate | Moderate |
| Key Finding | All grafted materials were more stable than the unmodified carbon. | Sample with lowest APTES showed highest stability. | More acidic surface groups on the carbon precursor led to more stable dispersions. |
Controlled Drug Release System
The primary amine groups on 3DOMC-APTES can form acid-labile imine bonds (C=N) with carbonyl groups of drug molecules, creating a pH-responsive release system. This is particularly useful for targeting the acidic microenvironment of tumor tissues [40].
Protocol: Drug Conjugation and In-Vitro Release Study
- Drug Loading: Conjugate a model drug (e.g., Nintedanib) to the 3DOMC-APTES carrier via the imine bond formation. This typically involves incubating the drug with the nanocarrier in a suitable solvent for 24-48 hours [40].
- Release Study:
- Place the drug-loaded nanocarrier in buffers at physiological pH (7.4) and acidic pH (5.5, mimicking the cancerous environment).
- Maintain the system at 37 °C under constant agitation.
- At predetermined time intervals, collect samples and analyze the drug concentration using UV-Vis spectroscopy or HPLC.
- Expected Outcome: A controlled and sustained release profile is observed. At pH 7.4, the release is prolonged, while at pH 5.5, the imine bond hydrolyzes rapidly, triggering a swift and extensive drug release (~85% over 48 hours) [40].
The thermodynamic parameters for such a system, determined by microcalorimetry, further elucidate the mechanism. The drug loading process is typically exothermic (ΔH ≈ -42.64 kJ mol⁻¹) and driven by enthalpy, indicating relatively weak interactions like hydrogen bonding. In contrast, the release process is endothermic (ΔH ≈ 10.30 kJ mol⁻¹), confirming that energy input (such as the lower pH in the tumor microenvironment) facilitates bond breakage and drug release [41].
Table 3: Quantitative Drug Release Profile of a Model System [40].
| Release Condition | Release Duration | Cumulative Drug Release | Key Mechanism |
|---|---|---|---|
| pH 7.4 (Physiological) | 48 hours | Prolonged, lower release | Slow hydrolysis of imine bond |
| pH 5.5 (Acidic) | 48 hours | ~85% | Acid-triggered rapid hydrolysis of imine bond |
| Microwave-Triggered | Multiple cycles | ~71% after 4 cycles | Thermal energy breaks hydrogen bonds [41] |
Visualization of the Drug Release Mechanism
The following diagram details the mechanism of pH-dependent drug release from the APTES-functionalized carrier.
Mechanism of pH-Triggered Drug Release
The integration of three-dimensionally (3D) ordered mesoporous carbons (OMCs) into pharmaceutical science represents a paradigm shift in drug delivery system (DDS) design. These materials, characterized by their high surface area, tunable pore architecture, and interconnected 3D networks, provide an ideal platform for enhancing drug loading and controlling release kinetics [22] [1]. This application note details specialized adsorption methodologies for loading two critical drug classes—antibiotics and poorly soluble drugs—into 3D OMCs, specifically CMK-8 and CMK-9. The protocols are framed within a broader thesis on the fabrication and functionalization of 3D OMCs, providing researchers with reproducible methods to leverage their unique structural advantages for advanced drug delivery applications.
Quantitative Performance of Mesoporous Carbon Drug Carriers
The following tables summarize key performance data for various mesoporous carbon carriers when loaded with different drug molecules, highlighting the impact of carbon architecture and surface chemistry.
Table 1: Adsorption and Release Performance for Antibiotic and Antihypertensive Drugs
| Mesoporous Carbon Type | Drug Loaded | Adsorption Capacity (mg/g) | Maximum Release (%) | Release Duration (h) | Key Release Condition |
|---|---|---|---|---|---|
| CMK-9 (Functionalized) | Cephalexin (Antibiotic) | 354 | 89% | 14 | pH 1.2 [22] |
| CMK-8 (Functionalized) | Cephalexin (Antibiotic) | 339 | Not Specified | 14 | pH 1.2 [22] |
| Urea-Modified CKIT-6 | Losartan Potassium (Antihypertensive) | 280 | 98.3% | Not Specified | pH 7.2 [42] |
| CTAB-Modified CKIT-6 | Losartan Potassium (Antihypertensive) | Not Specified | 98.3% | Not Specified | pH 7.2 [42] |
Table 2: Performance of Mesoporous Carbons for Poorly Soluble Drugs
| Mesoporous Carbon Type | Drug Loaded (BCS Class II) | Drug Loading Efficiency | Key Stability Finding | Reference |
|---|---|---|---|---|
| Hollow Mesoporous Carbon (HMC) | Indomethacin & Celecoxib | ~43% | Maintained dissolution profile for >12 months | [43] [44] |
| Mesoporous Carbon Nanoparticles (MCN) | Indomethacin & Celecoxib | Lower than HMC | Maintained dissolution profile for >12 months | [43] [44] |
Experimental Protocols for Drug Loading and Release
Functionalization of 3D OMCs (CMK-8 and CMK-9)
This step is critical for enhancing the aqueous dispersibility of the carbon carriers and introducing surface groups that can interact with drug molecules for controlled release [22].
- Objective: To graft 3-aminopropyltriethoxysilane (APTES) onto the surface of CMK-8 and CMK-9 to create amine-functionalized carriers.
Materials:
- Synthesized CMK-8 or CMK-9 carbon [22]
- 3-aminopropyltriethoxysilane (APTES)
- Toluene (anhydrous)
- Inert atmosphere (e.g., N₂ or Ar gas)
- Heating mantle with magnetic stirrer
- Soxhlet apparatus
- Ethanol
- Vacuum oven
Procedure:
- Drying: Activate the pristine CMK-8 or CMK-9 carbon by drying in a vacuum oven at 110°C for a minimum of 12 hours to remove adsorbed water.
- Dispersion: Under an inert atmosphere, disperse 1.0 g of the dried carbon in 100 mL of anhydrous toluene.
- Grafting: Add 4 mL of APTES to the dispersion. Heat the mixture to 80°C and reflux with constant stirring for 24 hours.
- Washing: After cooling, recover the functionalized carbon by filtration. Wash thoroughly with ethanol to remove any physisorbed silane.
- Purification: Further purify the product using a Soxhlet extractor with ethanol as the solvent for 24 hours.
- Drying: Dry the final functionalized product (denoted as C8-APTES or C9-APTES) in a vacuum oven at 60°C overnight [22].
Drug Loading via Solvent Evaporation Adsorption
This protocol is effective for both antibiotic molecules like cephalexin and poorly soluble drugs like indomethacin [22] [43].
- Objective: To load a high concentration of the drug into the mesopores of the functionalized OMC in an amorphous state.
Materials:
- Functionalized CMK-8 or CMK-9
- Drug (e.g., Cephalexin, Indomethacin, Celecoxib)
- Suitable solvent (e.g., water, methanol, ethanol)
- Ultrasonic bath
- Magnetic stirrer with hotplate
- Vacuum oven
- Centrifuge
Procedure:
- Drug Solution: Prepare a concentrated solution of the drug in a suitable solvent. For cephalexin, this may be water [22]. For poorly soluble drugs like indomethacin, use an organic solvent like methanol [43].
- Carrier Dispersion: Disperse a known mass (e.g., 100 mg) of the functionalized OMC in the drug solution. The typical carrier-to-drug ratio can start at 2:1.
- Incubation: Subject the mixture to ultrasonication for 30 minutes to ensure initial penetration of the drug solution into the pores.
- Solvent Evaporation: Stir the mixture continuously at 40°C while allowing the solvent to evaporate slowly over 24 hours.
- Drying: Once the solvent is fully evaporated, transfer the solid residue to a vacuum oven and dry at 40°C for 12 hours to remove any residual solvent [22] [43].
- (Optional) Washing: For poorly soluble drugs, a mild washing step may be incorporated to remove surface crystalline drug, leaving only the amorphous drug within the pores [43].
In Vitro Drug Release Studies
Evaluating the release profile is essential to confirm the controlled-release capability of the drug-loaded OMC system.
- Objective: To quantify the drug release kinetics from the loaded OMC under simulated physiological conditions.
Materials:
- Drug-loaded OMC powder
- Release media (e.g., simulated gastric fluid at pH 1.2; simulated intestinal fluid at pH 6.8)
- Water bath shaker
- Centrifuge tubes
- Centrifuge
- UV-Vis Spectrophotometer or HPLC system
Procedure:
- Incubation: Disperse an accurately weighed quantity of the drug-loaded OMC (e.g., 10 mg) in 50 mL of release medium pre-heated to 37°C.
- Agitation: Place the suspension in a water bath shaker maintained at 37°C and agitate at a constant speed (e.g., 100 rpm).
- Sampling: At predetermined time intervals, withdraw 1 mL of the release medium and immediately replace it with an equal volume of fresh, pre-warmed medium to maintain sink conditions.
- Analysis: Clarify the sample by centrifugation. Analyze the drug concentration in the supernatant using a validated analytical method, such as UV-Vis spectrophotometry at the λ_max of the drug or HPLC [22].
- Data Modeling: Fit the cumulative release data to kinetic models (e.g., Weibull model) to understand the release mechanism. A Fickian diffusion-controlled release is often observed for OMC-based systems [22].
Workflow and Mechanism Visualization
The following diagram illustrates the integrated experimental workflow for the functionalization, drug loading, and release testing of 3D ordered mesoporous carbons.
Figure 1: Experimental Workflow for OMC-Based Drug Delivery.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for OMC-Based Drug Loading Experiments
| Reagent / Material | Function / Role in the Protocol | Key Rationale |
|---|---|---|
| CMK-8 / CMK-9 Carbons | Primary drug carrier. CMK-9's dual mesoporosity offers superior loading capacity. | 3D cubic structure (Ia3d) with interconnected pores enhances molecular diffusion and confinement [22]. |
| 3-Aminopropyltriethoxysilane (APTES) | Surface functionalizing agent. | Introduces amine groups, improving aqueous dispersibility and enabling pH-responsive release via electrostatic interactions [22]. |
| Cephalexin | Model antibiotic drug. | Short half-life drug that benefits from controlled release, reducing dosing frequency [22]. |
| Indomethacin / Celecoxib | Model BCS Class II drugs (poorly soluble). | Demonstrate the ability of OMCs to amorphize and enhance the solubility and stability of insoluble drugs [43] [44]. |
| Cetyltrimethylammonium Bromide (CTAB) | Alternative surface modifier (surfactant). | Can alter surface charge and hydrophobicity, fine-tuning drug-carrier interactions for specific molecules like losartan [42]. |
| Anhydrous Toluene | Solvent for APTES functionalization. | Prevents premature hydrolysis of the silane agent during the grafting process [22]. |
Overcoming Synthesis and Performance Challenges in 3DOMC Fabrication
Addressing High Production Costs and Commercialization Barriers
The fabrication of three-dimensionally ordered mesoporous carbon (3DOMC) materials holds tremendous promise for applications ranging from energy storage to drug delivery. However, the transition from laboratory-scale synthesis to commercial implementation faces significant economic and technical hurdles. The complex production processes required to achieve precise pore structures demand specialized equipment and expertise, resulting in elevated operational and capital expenditures that directly translate to higher unit costs [45]. Furthermore, scaling manufacturing to meet industrial demands while maintaining structural integrity and pore characteristics presents considerable inefficiencies, restricting widespread adoption across key sectors [45]. This application note analyzes these barriers and provides detailed protocols for implementing more sustainable and cost-effective synthesis approaches.
Quantitative Analysis of Cost Drivers & Solutions
The economic viability of 3DOMC materials is influenced by multiple factors, from precursor selection to template removal processes. The table below summarizes the primary cost drivers and corresponding mitigation strategies supported by recent research.
Table 1: Key Cost Drivers and Economical Synthesis Strategies for 3D Ordered Mesoporous Carbons
| Cost Driver | Impact Level | Economical Solution | Reported Efficacy |
|---|---|---|---|
| Template Materials & Removal | High | Use of self-template techniques and biomass-derived precursors [46] [3]. | Reduces reliance on expensive mesoporous silica templates and hazardous etchants [46]. |
| Activation Agents | Medium | Exploration of green activators and safe chemical agents [46]. | Minimizes use of corrosive/hazardous chemicals, lowering purification needs and environmental impact [46]. |
| Carbon Precursors | Medium | Utilization of conjugated polymeric frameworks or biomass [47] [46]. | Phytic acid cross-linked framework yielded ~50 wt% carbon, >2x biomass carbonization [47]. |
| Scaling Inefficiencies | High | Simplified soft-templating and aqueous routes [48] [3]. | Aqueous pathway offers better reproducibility and unlimited batch size vs. EISA [48]. |
| Energy Consumption | Medium | Low-temperature graphitization (as low as 800°C) [47]. | Maintains high surface area (4073 m²/g) and hierarchical pores at reduced energy cost [47]. |
Detailed Experimental Protocols
Protocol 1: Sustainable Synthesis of Nitrogen-Doped Ordered Mesoporous Carbon (N-OMC)
This protocol outlines the synthesis of N-OMC using a covalent triazine framework as a dual carbon and nitrogen precursor, reducing the need for separate doping agents [49].
Materials & Equipment:
- Pluronic P123 (structure-directing agent)
- Tetraethylorthosilicate (TEOS, silica source)
- 1,4-Dicyanobenzene (carbon/nitrogen precursor)
- ZnCl₂ (catalyst)
- Hydrochloric acid (HCl, 37 wt%)
- Sodium hydroxide (NaOH, 1.0 M)
- Acetone (solvent)
- Autoclave
- Tube furnace
- Soxhlet extractor
Procedure:
- SBA-15 Silica Template Synthesis:
- Dissolve 4.0 g Pluronic P123 in 126 mL deionized water with stirring.
- Add 20 mL HCl (37 wt%) and 9.2 mL TEOS sequentially. Stir for 20 h at 35°C.
- Transfer solution to autoclave for hydrothermal treatment at 100°C for 12 h.
- Filter, dry, and calcine at 550°C for 6 h to remove the template [49].
Covalent Triazine Framework (CTF) Preparation:
- Mix 1,4-dicyanobenzene and ZnCl₂ in a 1:1 molar ratio.
- Heat mixture at 400°C for 40 h in an inert atmosphere.
- Cool to room temperature, wash with dilute HCl to remove ZnCl₂, and dry [49].
Nanocomposite Formation and Carbonization:
- Mix 2 g CTF and 1 g SBA-15 in 30 mL acetone. Reflux with stirring for 3 h.
- Centrifuge mixture and dry at 50°C.
- Transfer powder to porcelain boat and carbonize at 800°C for 2 h under inert gas (N₂/Ar) with a heating rate of 5°C/min [49].
Template Removal:
- Treat silica-carbon nanocomposite with 1.0 M NaOH solution in a 100°C autoclave for 6 h.
- Repeat treatment twice to ensure complete silica removal [49].
Characterization & Expected Outcomes:
- Surface Area: ~1130 m²/g (comparable to CMK-9 materials) [22]
- Nitrogen Content: ~7.5% (via CHN analysis) [49]
- Morphology: Fibrous structures with retained mesoporous order [49]
Protocol 2: Three-Dimensional OMC/CNT Nanocomposite for Enhanced Performance
This protocol creates a 3D conductive network by growing carbon nanotubes within an OMC framework, improving rate performance without costly post-synthesis modifications [50].
Materials & Equipment:
- Triblock copolymer F127 (soft template)
- Phenol-formaldehyde resol (carbon precursor)
- Nickel nitrate (Ni(NO₃)₂·6H₂O, catalyst precursor)
- Tetraethyl orthosilicate (TEOS, silica source)
- C₂H₂ gas (carbon source for CNTs)
- Ethanol, HCl, HF
- Chemical vapor deposition (CVD) system
Procedure:
- OMC/Ni Nanocomposite Synthesis:
- Dissolve 1.6 g F127 and 1.0 g 0.2 M HCl in 8.0 g ethanol.
- Add 0.13 g Ni(NO₃)₂·6H₂O and stir for 1 h at 40°C.
- Add 2.08 g phenol-formaldehyde resol (20 wt% in ethanol) and 4.16 g TEOS with stirring.
- Pour mixture into dishes, evaporate at room temperature for 8 h, then at 100°C for 24 h.
- Carbonize at 900°C under nitrogen. Remove silica with HF solution [50].
- In Situ CNT Growth via CVD:
- Place OMC/Ni nanocomposite in CVD furnace.
- Heat to 700°C under nitrogen flow.
- Introduce C₂H₂ gas for 5 minutes for CNT growth, using embedded Ni nanoparticles as catalysts.
- Cool to room temperature under nitrogen [50].
Characterization & Expected Outcomes:
- Specific Capacitance: 338.1 F/g at 1 A/g in 6 M KOH
- Rate Capability: 130.2 F/g at 50 A/g
- Cycling Stability: 91.6% capacity retention after 4000 cycles [50]
- Surface Area: 1197.2 m²/g with uniform pore distribution (~6.4 nm and 2.2 nm) [50]
Synthesis Workflow for 3D Ordered Mesoporous Carbons
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents for 3DOMC Synthesis and Their Functions
| Reagent/Chemical | Function in Synthesis | Cost-Saving/Sustainable Alternatives |
|---|---|---|
| Pluronic P123/F127 | Soft template for mesostructure control [48] [3] | Biomass-derived surfactants; optimized recycling protocols |
| SBA-15/KIT-6 Silica | Hard template for precise pore replication [22] [49] | Reusable templates; silica-free self-assembly routes [48] |
| Phenol-formaldehyde Resol | Carbon precursor for framework formation [50] [51] | Lignin or biomass extracts; covalent triazine frameworks [49] |
| Phytic Acid | Cross-linker and porogen in polymer frameworks [47] | Plant-derived phytic acid; alternative green cross-linkers |
| Nickel Nitrate | Catalyst precursor for in-situ CNT growth [50] | Catalyst recovery systems; non-noble metal alternatives |
| Hydrofluoric Acid (HF) | Silica template etching agent [50] [51] | Alkaline dissolution (NaOH/KOH) [49]; template recycling |
| Potassium Hydroxide (KOH) | Chemical activating agent [47] | Green activators (K₂CO₃, NaOH); physical activation methods [46] |
Advancing 3DOMC materials from research laboratories to commercial applications requires continued focus on sustainable precursor sources, simplified synthesis workflows, and scalable manufacturing protocols. The strategies outlined herein—including self-template techniques, low-temperature graphitization, and green activation methods—provide a viable pathway to overcome current cost barriers. Future research should prioritize template recycling protocols, continuous manufacturing systems, and application-specific structural optimization to further enhance the economic feasibility of these versatile materials.
Preventing Mesostructure Collapse During High-Temperature Treatments and Nitride Conversion
The fabrication of three-dimensionally ordered mesoporous carbon (3DOMC) materials represents a significant advancement in nanotechnology due to their uniform pore size, high specific surface area, and exceptional physicochemical properties. These materials have demonstrated remarkable potential in applications ranging from energy storage and conversion to catalytic supports and drug delivery systems [3]. However, a critical challenge persists in maintaining structural integrity during high-temperature processes essential for material synthesis and functionalization, particularly during nitride conversion reactions.
Mesostructure collapse typically occurs when the capillary forces exerted during precursor decomposition exceed the mechanical strength of the nascent porous framework. This phenomenon becomes especially pronounced during thermal treatments above 500°C and nitride conversion processes requiring ammonia exposure at elevated temperatures [52] [53]. The preservation of mesoscopic order during these processes is paramount for ensuring optimal performance in final applications, as collapsed structures exhibit diminished surface area, compromised pore accessibility, and reduced active site availability [3].
This Application Note provides a comprehensive framework of strategies and protocols to prevent mesostructure collapse, with particular emphasis on template reinforcement, thermal parameter optimization, and conversion pathway engineering. The protocols outlined herein are designed to integrate seamlessly within broader research initiatives focused on advancing 3DOMC-based material systems for scientific and industrial applications.
Fundamental Challenges and Stabilization Mechanisms
Primary Causes of Mesostructure Collapse
The degradation of 3DOMC architectures during high-temperature processing stems from multiple interrelated factors:
- Thermal Sintering: Carbon framework shrinkage and pore coalescence occur at elevated temperatures as surface energy minimization drives structural reorganization [52]. This process is exacerbated in materials with thin pore walls and low degrees of graphitization.
- Capillary Stress: During precursor decomposition and template removal, the liquid-to-gas phase transition generates substantial capillary forces that can exceed the mechanical strength of the mesoporous framework [3].
- Structural Failure During Nitridation: The conversion of metal oxide precursors to nitrides under ammonia flow involves significant molar volume changes and crystalline phase transformations that introduce substantial mechanical stress [53] [54]. The replacement of oxygen with nitrogen atoms within the metal lattice expands the unit cell volume, potentially fracturing the delicate carbon scaffold.
Stabilization Strategies
Several complementary approaches have demonstrated efficacy in preserving mesostructure integrity:
- Rigid Template Confinement: Using robust templates such as silica colloidal crystals or mesoporous silica during carbonization provides physical confinement that restricts framework shrinkage [38] [3].
- Carbon Scaffold Reinforcement: Introducing a secondary carbon source or increasing the carbon precursor concentration strengthens the pore walls, enhancing resistance to thermal degradation [53].
- Optimized Thermal Programming: Controlled heating rates and isothermal holding periods at critical temperatures allow gradual stress relaxation and prevent rapid gas evolution that can damage the porous network [52] [54].
Table 1: Quantitative Comparison of Mesoporous Carbon Stability Under High-Temperature Conditions
| Material System | Stabilization Approach | Maximum Temperature (°C) | Structural Outcome | Specific Surface Area Retention |
|---|---|---|---|---|
| 3DOMC (baseline) | None | 450 | Partial collapse | ~60% |
| Ni/3DOMC [38] | Confined growth of Ni nanosheets | 850 | Maintained mesostructure | >85% |
| ZnO/OMCS [39] | ZnO nanoparticle reinforcement | 900 | Maintained spherical morphology | >90% |
| TiNbN [53] | Carbon scaffold retention | 600 | Preserved gyroidal structure | ~80% |
| OMC (hard template) [52] | Silica template confinement | 1000 | Ordered structure maintained | 75-85% |
Experimental Protocols
Protocol 1: Hard-Templated 3DOMC Synthesis with Enhanced Thermal Stability
This protocol describes the synthesis of 3DOMC using silica colloidal crystal templates with modifications to enhance high-temperature stability, adapted from established methodologies with critical improvements [38] [39] [3].
Research Reagent Solutions
Table 2: Essential Reagents for 3DOMC Synthesis
| Reagent | Function | Specifications | Alternative Options |
|---|---|---|---|
| PMMA colloidal crystals | Macroporous template | Sphere diameter: 300-400 nm | PS colloidal crystals |
| Furfuryl alcohol | Carbon precursor | ≥98% purity | Sucrose, phenolic resin |
| Silica inverse opal | Mesoporous template | Pore size: 10-80 nm | SBA-15, MCM-48 |
| Oxalic acid | Polymerization catalyst | ≥99% purity | p-Toluenesulfonic acid |
| Ethanol | Solvent | Anhydrous | Methanol, isopropanol |
| Hydrofluoric acid | Silica etching | 5% solution | NaOH/KOH (for some templates) |
Step-by-Step Procedure
Silica Inverse Opal Template Preparation
- Immerse PMMA colloidal crystal template (1×1 cm) in silica precursor solution (TEOS:EtOH:0.1M HCl = 1:1:1.5 mass ratio) for 60 minutes under ambient conditions.
- Remove impregnated template and dry at room temperature for 12 hours.
- Calcinate at 450°C for 5 hours (heating rate: 1°C/min) to remove PMMA template, resulting in silica inverse opal.
Carbon Precursor Infiltration
- Prepare carbon precursor solution: 1g phenolic resol and 1g F127 triblock copolymer in 20mL ethanol.
- Immerse silica inverse opal template in precursor solution for 120 minutes.
- Remove template and evaporate solvent at room temperature for 24 hours.
Controlled Carbonization
- Place precursor-loaded template in tube furnace under nitrogen atmosphere.
- Program thermal treatment:
- Ramp 1°C/min to 350°C, hold for 5 hours (F127 removal)
- Ramp 5°C/min to 900°C, hold for 2 hours (carbonization)
- Cool naturally to room temperature under continuous N2 flow.
Template Removal
- Immerse carbon/silica composite in 5% HF solution for 72 hours to etch silica template.
- Wash resulting 3DOMC with deionized water (3×) and ethanol (1×).
- Dry at 80°C under vacuum for 12 hours.
Critical Parameters for Mesostructure Preservation
- Heating Rate Control: The initial slow heating rate (1°C/min) during template removal prevents rapid gas evolution that causes pore collapse.
- Atmosphere Control: Continuous inert gas flow prevents oxidative degradation of the carbon framework.
- Gradual Template Etching: Controlled HF concentration and extended etching time prevent mechanical damage to the fragile carbon network.
Protocol 2: Nitride Conversion with Structural Preservation
This protocol describes the conversion of metal oxide/3DOMC composites to transition metal nitrides while maintaining mesostructural integrity, based on successful demonstrations with mixed titanium-niobium systems [53] [54].
Research Reagent Solutions
Table 3: Essential Reagents for Nitride Conversion
| Reagent | Function | Specifications | Alternative Options |
|---|---|---|---|
| Ammonia gas | Nitriding agent | Anhydrous, ≥99.8% | N2/H2 mixture (25:75) |
| Metal oxide/3DOMC composite | Precursor material | Surface area >500 m²/g | Various transition metals |
| Urea | Alternative nitrogen source | ≥99% purity | Melamine, dicyandiamide |
Step-by-Step Procedure
Metal Oxide/3DOMC Composite Preparation
- Prepare metal precursor solution (e.g., 0.9g Zn(CH3COO)2·2H2O in 25mL methanol) [39].
- Disperse 0.5g 3DOMC in precursor solution via ultrasonication for 60 minutes.
- Add precipitating agent (0.45M KOH in methanol) dropwise under continuous stirring.
- Recover solid by filtration and dry at 70°C overnight.
Optimized Nitride Conversion
- Load metal oxide/3DOMC composite into quartz boat placed in tubular reactor.
- Purge system with argon (100mL/min) for 30 minutes.
- Initiate temperature program:
- Ramp 1°C/min to 300°C under argon
- Switch gas to ammonia (50mL/min)
- Ramp 0.5°C/min to target temperature (500-700°C, depending on metal)
- Hold for 2-4 hours
- Cool to room temperature under ammonia flow.
- Passivate surface if necessary with 1% O2/Ar mixture for 60 minutes.
Critical Parameters for Structural Preservation
- Controlled Heating Rates: The exceptionally slow heating during nitridation (0.5°C/min) allows gradual phase transformation without generating destructive stress.
- Gas Switching Protocol: Initial decomposition under inert atmosphere prevents premature reaction between carbon and ammonia.
- Temperature Optimization: Different metals require specific maximum temperatures (500-600°C for Ti-based, 600-700°C for Mo-based nitrides) to balance complete conversion against structural degradation [54].
Workflow Visualization
Mesoporous Material Stabilization Strategy
High-Temperature Nitride Conversion Workflow
Characterization and Quality Control
Structural Validation Methods
Rigorous characterization is essential to confirm mesostructure preservation following high-temperature treatments:
- Small-Angle X-Ray Scattering (SAXS): Monitors maintenance of mesoscale order through the presence of characteristic peaks corresponding to the mesoporous structure [53]. A quality 3DOMC material should exhibit at least three discernible peaks with consistent d-spacing ratios.
- N₂ Physisorption Analysis: Quantifies specific surface area (BET method) and pore size distribution (BJH method). Successful structure preservation demonstrates type-IV isotherms with sharp capillary condensation steps and minimal hysteresis [38] [3].
- Transmission Electron Microscopy: Provides direct visualization of pore ordering and wall integrity. High-quality samples show uniform periodic arrangements without regions of collapse or disorder [39].
Performance Metrics
Table 4: Quality Assessment Parameters for 3DOMC After High-Temperature Processing
| Parameter | Acceptance Criterion | Measurement Technique | Typical Optimal Values |
|---|---|---|---|
| Specific Surface Area | >80% retention vs. pre-treatment | N₂ physisorption | >800 m²/g |
| Pore Volume | >75% retention vs. pre-treatment | N₂ physisorption | >0.8 cm³/g |
| Mesopore Ordering | Maintained SAXS pattern | SAXS/XRD | Distinct (100), (110), (200) peaks |
| Wall Crystallinity | Appropriate for application | Raman spectroscopy | ID/IG ratio 0.8-1.2 |
| Mechanical Stability | Withstands processing stresses | Nanoindentation | Hardness >0.5 GPa |
Troubleshooting Guide
Common Issues and Solutions
Problem: Complete structural collapse during carbonization
Problem: Partial pore blocking after nitride conversion
Problem: Reduced surface area after high-temperature treatment
Problem: Phase inhomogeneity in mixed metal nitrides
The preservation of mesostructural integrity during high-temperature treatments and nitride conversion processes is achievable through meticulous control of synthesis parameters and implementation of strategic reinforcement approaches. The protocols outlined in this Application Note provide validated methodologies for maintaining structural order under demanding processing conditions, enabling the fabrication of advanced 3DOMC-based materials with enhanced performance characteristics.
Critical success factors include the use of rigid templates during carbonization, controlled thermal programming with gradual heating rates, and optimized nitridation protocols that minimize structural stress. When properly implemented, these approaches enable the production of mesoporous materials that retain high surface areas and ordered pore structures even after exposure to temperatures up to 900°C [38] [39].
These stabilization strategies open new possibilities for employing 3DOMC materials in high-temperature applications including catalysis, energy storage, and gas separation, where structural robustness is essential for long-term performance and commercial viability.
Optimizing Ink Formulation and Rheology for 3D Printing Stability
The fabrication of advanced materials, such as three-dimensionally ordered mesoporous carbon, increasingly relies on Direct Ink Writing (DIW) for its ability to create complex, architectured monoliths with tailored macro- and mesostructures [55]. The successful application of these materials in catalysis, sensing, and energy storage hinges on the printability and structural integrity of the printed scaffolds. Achieving this requires a meticulous balance between the ink's chemical composition and its rheological behavior, coupled with the optimization of printing parameters [56] [55]. These factors are deeply interrelated; a well-formulated ink can still fail if processed incorrectly, and optimal parameters are useless without an ink possessing the correct viscoelastic properties. This protocol provides a detailed, systematic framework for optimizing both ink formulation and the DIW process to ensure the production of stable, high-fidelity porous carbon structures, directly supporting research within the context of mesoporous carbon fabrication.
Workflow for Systematic Optimization
The following workflow outlines the key stages for developing and optimizing a DIW process, from initial ink formulation to final performance evaluation.
Systematic Optimization Workflow for DIW: This diagram outlines the iterative process for developing a stable and functional 3D printing ink, highlighting key experimental stages like rheological characterization and parameter optimization [57] [58].
Experimental Protocols
Protocol 1: Rheological Characterization of Inks
Objective: To quantitatively measure the key rheological properties that determine ink printability, including yield stress, shear-thinning behavior, and viscoelastic moduli [56] [59].
Sample Preparation:
- Prepare the ink formulation and ensure homogeneous mixing.
- Load the sample onto the rheometer plate, carefully trimming the excess to ensure a consistent geometry for measurement.
Yield Stress and Flow Curve Measurement:
- Perform a controlled shear stress ramp (e.g., from 1 Pa to 1000 Pa) or a shear rate ramp.
- Record the resulting viscosity and shear stress.
- Fit the data to the Herschel-Bulkley model (Equation 1) to extract the yield stress (σ_Y), consistency index (k), and flow index (n) [59].
- Equation 1 (Herschel-Bulkley):
σ = σ_Y + k * γ˙^n, whereσis shear stress andγ˙is shear rate.
Amplitude Sweep for Viscoelasticity:
- Conduct an oscillatory amplitude sweep at a fixed frequency (e.g., 1 Hz).
- Measure the storage modulus (G') and loss modulus (G'') as a function of strain.
- Determine the critical strain (the point where G' and G'' cross, indicating the solid-to-liquid transition) and the loss factor, tan(δ) (G''/G') at low strains [59].
Data Interpretation: Refer to Table 1 for target values that indicate a printable ink.
Protocol 2: A Two-Step Method for Optimizing DIW Process Parameters
Objective: To systematically identify the optimal printing parameters that ensure dimensional accuracy and shape fidelity of the printed structures [58].
Step 1: Optimize Parameters for Printing a Single Line This step focuses on achieving a continuous, uniform filament free from interruptions or excess material [56] [58].
- Screening Experiments: Perform preliminary tests to establish a "printability window" for each parameter.
- Taguchi Design of Experiments (DoE): Use an orthogonal array (e.g., L9) to efficiently test the interaction of key parameters.
- Key Parameters:
- Analysis: Calculate the Signal-to-Noise (S/N) ratio for the measured line width, with the target being the nozzle diameter. Use Analysis of Variance (ANOVA) to determine the significance of each parameter.
Step 2: Optimize the Parameter for Transition from Lines to a Layer This step ensures proper adhesion between adjacent filaments without deformation [58].
- Measure the average width of a line printed under the optimal conditions from Step 1.
- Conduct single-factor tests printing a solid rectangular object.
- Systemically adjust the road width or path spacing parameter in the slicing software. The optimal value is typically equal to or slightly less than the measured average line width.
Data Presentation and Analysis
Rheological Property Targets for DIW Inks
Table 1: Target ranges for key rheological properties to achieve a stable and printable DIW ink. These properties ensure extrudability, shape retention, and layer support [56] [59] [60].
| Property | Symbol | Target Range | Functional Significance in DIW |
|---|---|---|---|
| Yield Stress | σ_Y | ~500 Pa [58] to > ρgH [59] | Ensances shape retention after deposition and prevents slumping; must support the weight of subsequent layers. |
| Flow Index | n | < 1 (typically << 0.25 for strong shear-thinning) [59] | Enables shear-thinning: viscosity drops during extrusion for easy flow and recovers after deposition. |
| Loss Factor | tan(δ) | < 1 (G' > G'') at low strains [59] | Indicates solid-like, elastic behavior dominant at rest, crucial for shape fidelity. |
| Storage Modulus | G' | > Loss Modulus (G'') [58] | Provides elastic strength to resist deformation and support the structure after printing. |
Key Process Parameters and Optimization Strategy
Table 2: Key DIW process parameters and a summary of the optimization strategy to achieve dimensional accuracy [58].
| Parameter Category | Key Parameters | Optimization Method & Goal |
|---|---|---|
| Printing a Single Line | Nozzle Velocity (vn)Normalized Piston Ratio (RC)Normalized Height (H) | Method: Taguchi DoE with ANOVA.Goal: Achieve a continuous filament with a width matching the nozzle diameter. |
| Forming a Layer | Road Width / Path Spacing | Method: Single-factor tests based on optimal line width.Goal: Achieve complete, seamless infill without buckling or gaps. |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential materials and reagents for formulating and optimizing DIW inks for porous carbon structures.
| Reagent / Material | Function / Role | Example Application |
|---|---|---|
| Soy Protein Isolate (SPI) / Gelatin | Natural polymer binder and carbon source; provides biopolymeric network for the ink [55] [61]. | Used in starch/gelatin-based inks as a sustainable carbon precursor for porous carbon monoliths [55]. |
| Polyvinyl Alcohol (PVA) | Synthetic polymer additive; enhances mechanical strength and flexibility of the printed green body [61]. | Added to SPI inks to improve printability and the mechanical properties of the final scaffold [61]. |
| Sodium Alginate (SA) | Polysaccharide viscosifier; promotes physical cross-linking via hydrogen bonds, improving ink stability [61]. | Incorporated into protein-based inks to enhance viscoelastic properties and shape fidelity [61]. |
| Monodispersed SiO₂ Spheres | Functional filler and hard template; acts as a viscosifier and porogen for creating meso/macroporous structures [55]. | Mixed with starch/gelatin ink to tailor pore sizes in the final carbon replica after etching [55]. |
| 1-Ethynyl-1-cyclohexanol (ETCH) | Curing inhibitor; controls the reaction kinetics in cross-linking inks, improving shelf-life and printability [62]. | Used in silicone-based DIW formulations to prevent premature curing and allow for reliable printing [62]. |
| Carrageenan | Thermo-gelling agent; induces rapid solidification of the ink upon temperature change, aiding shape retention [60]. | Added to ceramic slurries for thermal-induced solidification after deposition in DIW [60]. |
Advanced Optimization and Analysis Techniques
Statistical and Machine Learning Approaches
For complex systems with multiple interacting variables, advanced statistical and computational methods can significantly enhance the optimization process.
- Full Factorial Analysis (FFA): As demonstrated in the "nano-FFA" approach for multiphoton 3D printing, FFA can be a crucial tool for systematically disentangling and optimizing the interrelated effects of chemical composition (e.g., photoinitiator concentration, crosslinker type) and process parameters (e.g., laser power) [57]. This method allows for the identification of key parameters and their interactions, providing a rational basis for ink development.
- Machine Learning (ML) and Transfer Learning (TL): These methods can quantitatively evaluate the contribution of each printing parameter to the final quality. For instance, a Transfer Learning-enhanced Gradient-Boosting Decision Tree (GBDT) model has been successfully used to predict surface quality in 3D printing, identifying layer height as the most critical parameter [63]. These approaches offer improved generalization and can lower the requirement for extremely large, costly experimental datasets [63].
Logical Framework for Ink Design and Failure Analysis
The following diagram provides a logical framework for designing an ink formulation and troubleshooting common printing failures, linking specific issues back to their potential root causes in rheology or processing.
Ink Design and Failure Analysis Logic: This chart connects target rheological behaviors to successful printing and helps diagnose the root causes of common failures, such as slumping (insufficient yield stress) or extrusion difficulty (inadequate shear-thinning) [56] [59].
Balancing Pore Size, Graphitization, and Mass Transport Limitations
Three-dimensionally ordered mesoporous carbon (3DOMC) materials represent a significant advancement in nanostructured materials, characterized by their highly ordered pore networks with pore sizes ranging from 2 to 50 nm. These materials combine several exceptional properties, including high specific surface area, tunable pore architecture, excellent electrical conductivity, and outstanding chemical/thermal stability [3] [16]. The interconnected 3D porous networks facilitate efficient mass transport of ions, molecules, and electrons, making them particularly valuable for applications requiring high kinetics and large surface areas, such as energy storage systems, drug delivery platforms, and catalytic supports [64] [22] [46].
The fundamental challenge in 3DOMC design lies in optimizing three often competing material characteristics: pore size distribution, graphitization degree, and mass transport efficiency. Larger pores enhance molecular accessibility but reduce volumetric capacity and can compromise mechanical stability. Higher graphitization improves electrical conductivity but typically requires higher synthesis temperatures that may collapse delicate pore structures. This application note provides a systematic framework for balancing these parameters through controlled synthesis protocols and characterization methodologies, enabling researchers to tailor 3DOMC materials for specific application requirements.
Synthesis Methodologies and Experimental Protocols
Hard Templating (Nanocasting) Approach
The hard templating method, also known as nanocasting, remains the most precise technique for creating 3DOMC with well-defined pore structures. This approach uses rigid solid templates (typically mesoporous silica) with the desired mesostructure, which is subsequently removed after carbon precursor infiltration and carbonization [16].
Protocol: Silica-Templated 3DOMC Synthesis
Template Preparation: Synthesize SiO₂ nanospheres (approximately 300 nm diameter) via the modified Stöber method [64].
- Add 10 mL ammonia (25 wt%) to a mixture of deionized water (20 mL) and ethanol (70 mL) under magnetic stirring (Solution I).
- Prepare Solution II by adding 10 mL tetraethylorthosilicate (TEOS) into 90 mL ethanol.
- Quickly add Solution II to Solution I with continuous stirring for 6 hours at room temperature.
- Recover SiO₂ nanospheres by centrifugation, wash with ethanol, and dry at 60°C.
Precursor Infiltration:
- Mix SiO₂ template with soluble starch (carbon source) in aqueous solution at controlled ratios.
- Evaporate water completely with continuous stirring at 80°C.
- Grind the composite into fine powder for homogeneous distribution.
Carbonization:
- Heat the template-precursor composite under inert atmosphere (N₂ or Ar).
- Use a programmed temperature ramp: 1°C/min to 350°C (hold 2 hours), then 5°C/min to 900°C (hold 2 hours) [64].
Template Removal:
Key Parameters:
- Template : Carbon precursor ratio controls final pore volume and wall thickness.
- Carbonization temperature influences degree of graphitization.
- Template removal method affects surface chemistry and defect concentration.
Protocol: CMK-8 and CMK-9 Synthesis from KIT-6 Silica Template
CMK-8 Synthesis [22]:
- Use KIT-6 silica as hard template with sucrose as carbon source and H₂SO₄ as catalyst.
- Slowly disperse 1 g KIT-6 in solution containing 1.86 g sucrose, 0.21 g H₂SO₄, and 7.3 g H₂O.
- Stir at room temperature for 3 hours to ensure adequate impregnation.
- Heat mixture at 100°C for 6 hours, then 160°C for additional 6 hours.
- Repeat impregnation step to enhance pore filling.
- Carbonize at 900°C for 2 hours under inert atmosphere.
- Remove silica template with NaOH or HF solution.
CMK-9 Synthesis [22]:
- Use furfuryl alcohol as carbon precursor with KIT-6 silica template.
- Polymerize furfuryl alcohol inside template channels.
- Follow similar carbonization and template removal steps as CMK-8 protocol.
Soft Templating Approach
Soft templating employs amphiphilic block copolymers that self-assemble with carbon precursors to create ordered mesostructures through cooperative assembly [28].
Protocol: Polymer-Templated 3DOMC Film Formation
Template Selection: Use amphiphilic block copolymers such as Pluronic F127 (EO₁₀₆PO₇₀EO₁₀₆), PEO-PPO-PEO triblock copolymers, or PS-b-PAN diblock copolymers [28].
Precursor Solution Preparation:
- Dissolve template polymer and carbon precursor (e.g., phenolic resol, furfuryl alcohol) in suitable solvent (ethanol, THF, or water).
- Typical mass ratio: 1-5% template, 10-20% carbon precursor, balance solvent.
Film Casting and Self-Assembly:
- Deposit solution on substrate via dip-coating, spin-coating, or doctor blading.
- Control evaporation rate and humidity to facilitate mesophase formation.
- Age film at controlled temperature (25-50°C) to complete self-assembly.
Thermal Processing:
- Use stepwise thermal treatment: 100-200°C to crosslink resin, then 350°C to degrade template, finally 600-900°C for carbonization under inert gas.
- Heating rates critical to prevent structural collapse: typically 1°C/min below 300°C, 2-5°C/min above 300°C.
Template-Free Self-Assembly
Template-free methods create mesoporosity through direct carbonization of designed precursors or phase separation mechanisms, offering simplified processing but less structural control [46] [16].
Protocol: Conjugated Polymer Framework Approach
Precursor Synthesis: Create cross-linked conjugated polymeric molecular framework using polyaniline (PANi) backbone with phytic acid cross-linker [47].
Gel Formation:
- Form hydrogel network by mixing aniline monomer, oxidizing agent, and phytic acid cross-linker.
- Convert hydrogel to aerogel via freeze-drying to maintain macroscopic structure.
Carbonization and Activation:
- Carbonize polymer aerogel at 400-900°C under inert atmosphere.
- For chemical activation, mix carbonized material with KOH (typical ratio 2:1-4:1 KOH:C).
- Heat treatment at 800°C for 1-2 hours under nitrogen flow.
- Wash thoroughly with dilute HCl and water to remove inorganic residues.
Property Control and Optimization Strategies
Pore Size and Architecture Engineering
Pore size distribution directly influences molecular accessibility and transport kinetics. The following table summarizes control strategies for pore architecture:
Table 1: Pore Size Control Strategies in 3DOMC Synthesis
| Control Method | Typical Pore Size Range | Mechanism | Applications |
|---|---|---|---|
| Template Size Selection | 5-50 nm | Using silica spheres/nanoparticles of defined diameter as template [64] | Size-selective adsorption, macromolecule delivery |
| Block Copolymer Template | 4-30 nm | Varying hydrophobic block length in amphiphilic copolymers (e.g., Pluronics) [28] | Controlled drug release, catalyst supports |
| Carbon Precursor Ratio | 2-15 nm | Adjusting wall thickness through precursor:template ratio [64] | Battery electrodes, capacitive storage |
| Activation Process | 1-5 nm (additional micropores) | Creating microporosity within mesoporous walls via KOH, CO₂, or steam activation [47] | Enhanced gas adsorption, supercapacitors |
Graphitization Enhancement
Graphitization degree critically determines electrical and thermal conductivity, chemical stability, and mechanical strength. Higher graphitization typically requires elevated temperatures (>800°C) but may compromise surface area and pore structure.
Table 2: Graphitization Enhancement Methods for 3DOMC
| Method | Process Conditions | Graphitization Outcomes | Impact on Properties |
|---|---|---|---|
| High-Temperature Treatment | 800-1200°C in inert atmosphere | Increased sp² carbon content, enhanced crystallinity [64] | Improved conductivity (>3× vs activated carbon), enhanced stability [47] |
| Catalytic Graphitization | Addition of transition metals (Fe, Ni, Co) during carbonization | Lower graphitization temperature, formation of graphitic domains | Maintained porosity with higher conductivity, potential metal contamination |
| Conjugated Polymer Precursors | Use of polyaniline, polypyrrole, or polyimide precursors | Enhanced inherent graphitic character at lower temperatures | High conductivity with maintained surface area (up to 4073 m²/g) [47] |
| Heteroatom Doping | Introduction of N, B, or P during synthesis | Modified electronic structure without high temperature | Surface functionality enhancement, catalytic activity improvement [49] |
Surface Chemistry Modification
Surface functionalization expands 3DOMC application potential by introducing specific chemical interactions while maintaining structural integrity.
Protocol: Aminofunctionalization of 3DOMC
- Materials: 3DOMC, 3-aminopropyltriethoxysilane (APTES), anhydrous toluene.
- Procedure:
- Dry 3DOMC at 120°C for 12 hours under vacuum to remove adsorbed water.
- Dispense 1 g dried 3DOMC in 50 mL anhydrous toluene.
- Add 2 mL APTES dropwise with continuous stirring under nitrogen atmosphere.
- Reflux mixture at 110°C for 24 hours with stirring.
- Cool to room temperature, recover functionalized 3DOMC by filtration.
- Wash thoroughly with toluene and ethanol to remove unreacted silane.
- Dry at 80°C for 6 hours [22].
Materials and Characterization Data
Research Reagent Solutions
Table 3: Essential Research Reagents for 3DOMC Synthesis
| Reagent Category | Specific Examples | Function | Considerations |
|---|---|---|---|
| Hard Templates | SiO₂ nanospheres, KIT-6, SBA-15 [64] [22] [49] | Structure-directing scaffold for mesoporosity | Particle size determines pore diameter; surface chemistry affects precursor infiltration |
| Soft Templates | Pluronic F127, P123, Brij series [28] | Self-assembling mesophase directors | Hydrophilic-lipophilic balance controls mesostructure symmetry |
| Carbon Precursors | Sucrose, furfuryl alcohol, phenolic resol, polyaniline, starch [64] [22] [47] | Carbon source forming the framework | Influces carbon yield, heteroatom content, and graphitization tendency |
| Cross-linkers | Phytic acid, formaldehyde, hexamine [47] | Stabilizes polymer network before carbonization | Affects pore volume and structural integrity during pyrolysis |
| Activation Agents | KOH, NaOH, CO₂, steam [47] [46] | Creates additional microporosity and increases surface area | Concentration and temperature control activation degree and pore size distribution |
| Doping Precursors | Melamine, dicyandiamide, boric acid [49] | Introduces heteroatoms for modified surface chemistry | Alters electronic properties and creates active sites |
Comparative Properties of 3DOMC Materials
Table 4: Characteristic Properties of Different 3DOMC Structures
| Material Type | Surface Area (m²/g) | Pore Volume (cm³/g) | Pore Size (nm) | Conductivity | Key Applications |
|---|---|---|---|---|---|
| PCBNs | 447.4 | 1.567 | 10-30 (hollow spheres) | High graphitization | Li-S batteries (760 mAh/g at 0.1C) [64] |
| CMK-8 | ~1000 | ~1.2 | 3-6 (interconnected) | Moderate | Drug delivery (339 mg/g cephalexin) [22] |
| CMK-9 | 1130 | ~1.4 | 3-8 (dual mesoporosity) | Moderate | Enhanced drug delivery (354 mg/g cephalexin) [22] |
| HPG Carbon | 4073 | 2.26 | Hierarchical (micro-meso) | Very high (3× AC) | Supercapacitors, Li-S batteries [47] |
| N-doped OMC | ~500-1000 | ~0.8-1.5 | 3-10 | Moderate-high | Biodiesel production (94% yield) [49] |
| ZnO/OMCS | ~400-800 | ~0.5-1.0 | 10-50 (ordered spheres) | Moderate | Microwave absorption (-39.3 dB at 10.4 GHz) [7] |
Application Performance Data
Energy Storage Applications
Table 5: 3DOMC Performance in Energy Storage Systems
| Application | Material Structure | Key Performance Metrics | Structure-Function Relationship |
|---|---|---|---|
| Lithium-Sulfur Batteries | 3D ordered porous carbon bulk networks (PCBNs) | 760 mAh/g after 150 cycles at 0.1C; 455 mAh/g after 400 cycles at 1C [64] | Hierarchical microstructure accommodates sulfur expansion, confines polysulfides, enhances kinetics |
| Supercapacitors | Hierarchical porous graphitic (HPG) carbon | Unprecedented energy storage capacity and rate capability; high stability [47] | Ultrahigh surface area (4073 m²/g) with hierarchical pores enables high ion adsorption/desorption |
| Lithium-Ion Batteries | 3DOMC with metal oxide nanoparticles | Improved cycling stability and rate capability [3] | Ordered mesopores facilitate ion transport, buffer volume changes |
Drug Delivery Applications
Table 6: 3DOMC Performance in Pharmaceutical Applications
| Drug/Carrier System | Loading Capacity | Release Profile | Key Advantages |
|---|---|---|---|
| Cephalexin/CMK-9 | 354 mg/g [22] | 89% release within 14 h at pH 1.2 [22] | Dual mesoporosity enables high loading and controlled release |
| Cephalexin/CMK-8 | 339 mg/g [22] | Controlled release over 14 h | 3D cubic structure with interconnected pores facilitates diffusion |
| General Drug Delivery | High loading due to large surface area and pore volume [16] | Controlled release via surface functionalization [16] | Surface modification enables targeted delivery and smart release mechanisms |
Experimental Workflows and Optimization Pathways
The synthesis and optimization of 3DOMC materials involves critical decision points that collectively determine the final material properties. The following diagram illustrates the key synthesis pathways and their influence on final material characteristics:
Diagram 1: 3DOMC Synthesis and Optimization Pathways. This workflow illustrates the interconnected relationships between synthesis methods, parameter optimization, resulting material properties, and target applications. Hard templating provides precise pore control, soft templating enables surface chemistry tuning, and template-free methods offer graphitization advantages.
Troubleshooting and Optimization Guidelines
Common Synthesis Challenges and Solutions
Structural Collapse During Carbonization
- Cause: Excessive heating rates or insufficient cross-linking of carbon precursor.
- Solution: Implement graded thermal profile (1-2°C/min through polymer degradation temperature range 300-500°C); enhance precursor cross-linking with phytic acid or similar multifunctional cross-linkers [47].
Poor Template Replication
Inadequate Graphitization
- Cause: Insufficient carbonization temperature or non-graphitizing carbon precursor.
- Solution: Increase final carbonization temperature (800-1200°C); use graphitizable precursors (polyaniline, pitch); incorporate catalytic graphitization agents (Fe, Ni salts) [47].
Pore Blockage After Functionalization
- Cause: Excessive surface modification or improper reaction conditions.
- Solution: Control functionalization reagent concentration; use milder reaction conditions; ensure adequate washing after modification [22].
Characterization Techniques for 3DOMC Materials
Comprehensive characterization is essential for correlating synthesis parameters with resulting material properties:
- Structural Analysis: XRD for mesostructure ordering and graphitic character; Raman spectroscopy for defect density (ID/IG ratio) [49].
- Texture Properties: N₂ physisorption at 77K for surface area (BET), pore volume, and pore size distribution (NLDFT/BJH methods) [64] [47].
- Surface Chemistry: XPS for elemental composition and functional groups; FTIR for chemical bonding [7] [49].
- Morphology: SEM for macroscopic morphology; TEM for nanoscale structure and pore ordering [64] [7].
- Thermal Stability: TGA for decomposition profiles and heteroatom content [49].
The rational design of three-dimensionally ordered mesoporous carbon materials requires careful balancing of pore architecture, graphitization degree, and surface functionality. Hard templating approaches provide exceptional control over pore size and ordering, while soft templating offers more flexible processing options. Template-free methods present advantages for specific applications where simplified synthesis is prioritized. Through systematic optimization of synthesis parameters—including template selection, carbon precursor chemistry, thermal treatment profiles, and post-synthetic modifications—researchers can tailor 3DOMC materials for specific application requirements across energy storage, drug delivery, catalysis, and sensing domains. The protocols and data provided in this application note establish a foundation for developing next-generation 3DOMC materials with enhanced performance characteristics.
Strategies for Improving Chemical and Structural Stability Under Operational Conditions
Within the broader context of advancing the fabrication of three-dimensionally ordered mesoporous carbon (3DOMC) materials, ensuring their chemical and structural stability under operational conditions represents a critical research frontier. These materials, characterized by pore sizes between 2 and 50 nm and a highly ordered architecture, offer significant advantages for applications in energy storage, catalysis, and biomedicine, including high specific surface area, excellent electrical conductivity, and improved mass transport [3] [13]. However, their performance and longevity can be severely compromised by structural degradation, such as pore collapse, active site sintering, and carbon corrosion, particularly in harsh electrochemical or high-temperature environments [65] [66]. This application note details proven strategies and provides standardized protocols to enhance the operational stability of 3DOMC-based systems, enabling their reliable use in advanced technological applications.
Stabilization Strategies and Mechanisms
Surface Modification and Coating
Concealing the carbon surface from reactive intermediates is an effective method to mitigate corrosion. Atomic Layer Deposition (ALD) excels in this regard by creating uniform, conformal, and pinhole-free thin films on high-aspect-ratio 3DOMC structures.
- Metal Oxide Coatings: ALD-grown iron oxide (
FeO_x) has been demonstrated to significantly enhance the cyclability of 3DOMC cathodes in aggressiveLi-O₂battery environments. This stable oxide layer protects the carbon surface from detrimental side reactions with reduced oxygen species, such asLiO₂, without blocking the essential mesopores for mass transport [65]. - Mechanism: The coating functions as a physical barrier, preventing direct contact between the carbon framework and corrosive electrolytes or reaction intermediates, thereby suppressing parasitic reactions that lead to carbon oxidation and capacity fade.
Structural Optimization and Hierarchical Pore Design
Precise control over the material's architecture is paramount for withstanding mechanical stress induced by reaction processes.
- Mesopore Volume Enhancement: In potassium-ion batteries, a larger mesopore volume provides internal space to accommodate the huge volume expansion during (de)potassiation processes. This acts as a buffer, mitigating mechanical strain and preventing pulverization of the carbon electrode, which is a common cause of capacity decay [67].
- Hierarchical 3D Ordered Meso-Macroporous Structures: Constructing materials with interconnected macroporous cages framed by mesoporous walls (3DOMM) introduces a "structural cage effect." The macropores facilitate efficient diffusion of reactants and products, preventing pore blocking, while the mesopores provide a high surface area and a confinement effect that inhibits the agglomeration and sintering of active metal nanoparticles (e.g., Ni, Pd) during high-temperature reactions like dry reforming of methane (DRM) [66].
Heteroatom Doping and Functionalization
Incorporating heteroatoms like nitrogen into the carbon lattice modifies its electronic properties and enhances its intrinsic stability.
- Nitrogen Doping: The substitution of carbon atoms with nitrogen in the graphitic framework increases the electron density of the material. This functionalization can improve the interaction between the carbon support and anchored metal nanoparticles, leading to stronger metal-support interactions (SMSI). Stronger SMSI reduces Ostwald ripening and particle mobility, key drivers of nanoparticle sintering and catalyst deactivation [68] [49].
Table 1: Quantitative Impact of Structural Parameters on Electrochemical Stability
| Structural Parameter | Influence on Stability | Quantitative Performance Example | Key Mechanism |
|---|---|---|---|
| Mesopore Volume | Positive linear correlation with capacity retention in PIBs [67]. | Capacity of 263.9 mA h g⁻¹ after 1000 cycles at 1000 mA g⁻¹ [67]. | Accommodates volume expansion, provides adsorption sites. |
| Hierarchical 3DOMM Structure | Enhances carbon deposition resistance in DRM [66]. | ~90% CH₄ conversion maintained over 100 hours at 650°C [66]. | Facilitates mass transport, oxidizes carbon deposits, confines metal nanoparticles. |
| Surface Coating (FeOₓ ALD) | Enables long-term cycling in Li-O₂ batteries [65]. | Cyclability of >68 cycles with capacity >6000 mA h g⁻¹ [65]. | Protects carbon surface from reactive oxygen species. |
Detailed Experimental Protocols
Protocol: Atomic Layer Deposition of FeOₓ on 3DOMC
This protocol outlines the procedure for applying a conformal FeO_x coating onto a 3DOMC cathode to enhance its stability in Li-O₂ batteries [65].
1. Materials and Equipment:
- 3DOMC Substrate: Synthesized via hard-templating (e.g., using KIT-6 silica).
- ALD Precursors: Ferrocene (
Fe(C₅H₅)₂, ≥99%) and ozone (O₃). - Equipment: Hot-wall tubular ALD reactor, nitrogen (
N₂) gas supply, precursor heating ovens.
2. Step-by-Step Procedure:
1. Substrate Loading: Place the 3DOMC material in the center of the ALD reactor.
2. Reactor Conditioning: Purge the reactor with N₂ carrier gas and heat to the deposition temperature of 150°C.
3. ALD Cycle Execution:
- a. Fe Precursor Dose: Introduce the ferrocene precursor vapor into the reactor for a defined pulse time (e.g., 3 s).
- b. First N₂ Purge: Purge the reactor with N₂ for 60 s to remove non-reacted precursors and by-products.
- c. Co-reactant Dose: Introduce the O₃ co-reactant into the reactor for a defined pulse time (e.g., 3 s).
- d. Second N₂ Purge: Purge the reactor with N₂ for 60 s.
- e. Cycle Repetition: Repeat steps a-d for 50-100 cycles to achieve the desired film thickness.
3. Quality Control:
- The growth per cycle (GPC) should be monitored using a quartz crystal microbalance (QCM).
- The final material should be characterized by TEM to confirm the uniformity and conformality of the
FeO_xcoating.
Protocol: Fabrication of 3D Ordered Meso-Macroporous (3DOMM) Ni/Al₂O₃-C Composite
This protocol describes the synthesis of a hierarchical 3DOMM Ni/Al₂O₃ catalyst, where the carbon component can be incorporated via pyrolysis, showcasing a structure that combats sintering and carbon deposition [66].
1. Materials:
- Macroporous Template: Polymethyl methacrylate (PMMA) microspheres.
- Mesoporous Template: Pluronic F127.
- Alumina Source: Aluminum isopropoxide.
- Metal Precursor: Nickel(II) nitrate hexahydrate (
Ni(NO₃)₂·6H₂O). - Solvent: Ethanol.
2. Step-by-Step Procedure:
1. 3DOMM Al₂O₃ Support Synthesis (EISA-CCT Method):
- a. Dissolve 3.2 g F127 in ethanol.
- b. Add 3 g aluminum isopropoxide to the solution and stir thoroughly.
- c. Infiltrate the solution into a pre-assembled colloidal crystal template of PMMA microspheres.
- d. Allow for evaporation-induced self-assembly (EISA).
- e. Calcinate the composite at 400°C for 3 hours to remove the PMMA and F127 templates, forming the 3DOMM Al₂O₃ support.
2. Ni Nanoparticle Deposition (Incipient-Wetness Impregnation):
- a. Prepare an aqueous solution of Ni(NO₃)₂·6H₂O with a concentration calculated to yield a 15 wt% Ni loading on the final support.
- b. Slowly add the solution dropwise to the 3DOMM Al₂O₃ support until incipient wetness is achieved.
- c. Dry the impregnated material at 80°C for 12 hours.
- d. Calcinate the material in air at 500°C for 3 hours.
- e. Reduce the catalyst under a H₂/N₂ flow at 650°C for 2 hours to form metallic Ni nanoparticles.
3. Key Characterization:
- SEM/TEM: To confirm the hierarchical 3DOMM structure and Ni nanoparticle dispersion.
- XRD: To identify crystalline phases (
γ-Al₂O₃, metallic Ni). - N₂ Physisorption: To measure specific surface area, pore volume, and pore size distribution.
The Scientist's Toolkit
Table 2: Essential Research Reagents and Materials for 3DOMC Stabilization
| Reagent/Material | Function/Application | Key Consideration |
|---|---|---|
| Pluronic F127 | Soft template for mesopore structure direction during synthesis [66]. | The molecular weight and concentration dictate final mesopore size and ordering. |
| KIT-6 or SBA-15 Silica | Hard template for synthesizing highly ordered 3DOMC [3] [13]. | Enables precise replication of a 3D porous structure with high fidelity. |
| Ferrocene (Fe(C₅H₅)₂) | Precursor for ALD of stabilizing FeOₓ coatings [65]. |
Sublimation temperature and reactivity with ozone determine ALD window and growth rate. |
| Nickel Nitrate (Ni(NO₃)₂·6H₂O) | Metal precursor for loading active nanoparticles (e.g., for catalysis) [66]. | Concentration and impregnation method control final nanoparticle size and dispersion. |
| Nitrogen Gas (N₂) | Inert atmosphere for pyrolysis, carbonization, and ALD processes [65] [66]. | High purity (>99.999%) is critical to prevent oxidation during high-temperature steps. |
| PMMA Microspheres | Colloidal crystal template for creating 3D ordered macroporous structures [66]. | Monodispersity is essential for forming a highly ordered macroporous framework. |
Concluding Remarks
The strategic enhancement of chemical and structural stability in 3DOMC materials is achievable through a multi-faceted approach. As detailed in these protocols, surface engineering via ALD, intrinsic structural optimization via pore volume and hierarchical design, and electronic modification via heteroatom doping are all powerful, experimentally-validated methods. The choice of strategy depends on the specific operational conditions and degradation mechanisms of the target application. Integrating these strategies—for example, creating a hierarchical 3DOMM carbon support that is subsequently stabilized with an ALD coating—represents the cutting edge of developing robust, high-performance functional materials for the most demanding energy and chemical conversion technologies.
Evaluating Performance and Efficacy in Controlled Drug Release
The fabrication of three-dimensionally ordered mesoporous carbon (3DOMC) materials represents a significant advancement in the design of novel drug delivery systems (DDS). These materials possess remarkable textural properties, including high specific surface area, large pore volume, and hierarchical porosity, which make them highly adaptable for controlled release applications [22] [3]. Their chemical stability, biocompatibility, and facile functionalization capability further enhance their utility in biomedical applications [69].
Among the family of ordered mesoporous carbons, CMK-8 and CMK-9 are particularly noteworthy due to their three-dimensional cubic structures with Ia3d symmetry, derived from KIT-6 mesoporous silica templates [22]. This case study provides a detailed comparison of CMK-8 and CMK-9 as nanocarriers for the controlled release of cephalexin (CFX), a β-lactam antibiotic with a short half-life (~1.1 h) that necessitates frequent dosing in conventional therapy [22] [70]. The investigation focuses on their synthesis, functionalization, drug loading capacity, and release kinetics under physiologically relevant conditions.
Background and Significance
The Challenge of Cephalexin Delivery
Cephalexin is a widely used antibiotic for treating bacterial infections, but its short half-life necessitates administration every 6-8 hours to maintain effective therapeutic concentrations [70]. This frequent dosing leads to significant plasma fluctuations and increases the risk of developing bacterial resistance [22]. Controlled release formulations can mitigate these challenges by sustaining therapeutic drug levels over extended periods, thereby improving patient compliance and treatment efficacy.
Structural Advantages of 3D Mesoporous Carbons
The three-dimensional porous architecture of CMK-8 and CMK-9 offers significant advantages over one or two-dimensional mesoporous systems for drug delivery applications. Their interconnected pore networks facilitate enhanced molecular diffusion and accessibility, which can be strategically modulated based on the guest molecule and pore architecture [22]. While CMK-8 forms a three-dimensional network of interconnected branched rods with intertubular mesopores, CMK-9 exhibits dual mesoporosity, consisting of both intratubular mesopores within carbon nanotubes and intertubular mesopores created after silica template removal [22]. This structural distinction gives CMK-9 a superior surface area and potentially enhanced drug loading capacity.
Material Characteristics and Properties
Synthesis and Structural Properties
Both CMK-8 and CMK-9 were synthesized using the hard-template method with KIT-6 mesoporous silica as the template [22] [71]. CMK-8 was synthesized using sucrose as a carbon precursor, which infiltrates the KIT-6 template and forms a 3D network of interconnected branched rods upon carbonization [22]. In contrast, CMK-9 was synthesized from furfuryl alcohol, resulting in dual mesoporosity with both intratubular and intertubular mesopores [22].
The materials were functionalized with 3-aminopropyltriethoxysilane (APTES) via grafting to enhance aqueous dispersion and control drug release [22]. This surface modification introduces amino groups that influence drug-carrier interactions through electrostatic forces and hydrogen bonding [70].
Table 1: Structural Parameters of CMK-8 and CMK-9 Materials
| Parameter | CMK-8 | CMK-9 | Measurement Technique |
|---|---|---|---|
| Structure | Cubic Ia3d | Cubic Ia3d | XRD, TEM |
| Surface Area | >500 m²/g [71] | 1130 m²/g [22] | BET Analysis |
| Pore Diameter | 3.2-6.6 nm [71] | Dual mesoporosity [22] | BJH Method |
| Total Pore Volume | 0.7-1.1 cm³/g [71] | Higher than CMK-8 [22] | BET Analysis |
| Pore Geometry | Intertubular mesopores [22] | Intra- and intertubular mesopores [22] | TEM, SAXS |
Morphological Characterization
SEM imaging revealed that both CMK-8 and CMK-9 exhibit similar morphologies with well-defined tortuous rod-like shapes [22]. The average particle sizes were estimated at 95 nm for CMK-8 and 85 nm for CMK-9 [22]. This morphology and particle size did not change significantly after drug functionalization and loading in both materials, consistent with previous observations for KIT-6 templates [22]. The tortuous morphology, combined with the nanoscale particle size, suggests favorable properties for molecular accessibility and diffusion within the materials.
Experimental Protocols
Synthesis of CMK-8 and CMK-9
Protocol 1: Synthesis of CMK-8 Carbon
- Slowly disperse 1 g of KIT-6 template in a solution containing 1.86 g of sucrose, 0.21 g of H₂SO₄ (catalyst), and 7.3 g of H₂O [22].
- Stir the mixture at room temperature for 3 hours to ensure adequate impregnation [22].
- Heat the mixture at 100°C for 6 hours, followed by a second heating at 160°C for an additional 6 hours [22].
- Carbonize the resulting silica/sucrose composite in a tubular furnace at 900°C for 6 hours under N₂ atmosphere [22].
- Remove the silica template by washing with 2M NaOH solution at 80°C for 24 hours [22].
- Recover the CMK-8 carbon by filtration, wash with distilled water, and dry at 120°C for 12 hours [22].
Protocol 2: Synthesis of CMK-9 Carbon
- Impregnate 1 g of KIT-6 template with a solution containing 1.5 mL of furfuryl alcohol and 0.03 g of oxalic acid (catalyst) [22].
- Polymerize the furfuryl alcohol within the template pores by heating at 85°C for 24 hours [22].
- Carbonize the resulting composite in a tubular furnace at 900°C for 6 hours under N₂ atmosphere [22].
- Remove the silica template by washing with 2M NaOH solution at 80°C for 24 hours [22].
- Recover the CMK-9 carbon by filtration, wash with distilled water, and dry at 120°C for 12 hours [22].
Surface Functionalization with APTES
Protocol 3: Amino-functionalization of Mesoporous Carbons
- Oxidize 1 g of the synthesized carbon material with 50 mL of 4M nitric acid at 50°C for 1 hour with stirring at 200 rpm [70].
- Wash the oxidized material with deionized water until the conductivity reaches 10 µS cm⁻¹ [70].
- Dry the material overnight at 120°C [70].
- React 1 g of the oxidized carbon with 50 mL of 3-aminopropyltriethoxysilane (APTES) in toluene (10% v/v) at 120°C for 24 hours under reflux and continuous stirring [70].
- Recover the functionalized material by filtration and wash with ethanol to remove unreacted APTES [70].
- Dry the final product (designated as C8-A for CMK-8 or C9-A for CMK-9) at 120°C for 12 hours [70].
Drug Loading and Release Studies
Protocol 4: Cephalexin Loading
- Prepare a cephalexin solution in ethanol at a concentration of 20 mg/mL [22].
- Add the carbon material (pristine or functionalized) to the drug solution at a ratio of 1:10 (w/v) [22].
- Stir the mixture for 24 hours at room temperature in the dark to ensure complete drug adsorption [22].
- Separate the drug-loaded carbon by centrifugation at 10,000 rpm for 10 minutes [22].
- Dry the resulting solid (designated as C8-F, C9-F, C8-AF, or C9-AF) under vacuum at 60°C for 12 hours [22].
- Determine the drug loading capacity by thermogravimetric analysis (TGA) or UV-Vis spectroscopy of the supernatant [22].
Protocol 5: In Vitro Release Kinetics
- Place 10 mg of drug-loaded material in a dialysis membrane bag (MWCO 12-14 kDa) [22].
- Immerse the bag in 200 mL of release medium (pH 1.2 for gastric conditions or pH 6.8 for intestinal conditions) at 37°C with constant stirring at 100 rpm [22].
- Withdraw 2 mL aliquots at predetermined time intervals (0, 0.5, 1, 2, 4, 6, 8, 10, 12, 14 hours) and replace with an equal volume of fresh medium to maintain sink conditions [22].
- Analyze the cephalexin concentration in the aliquots by HPLC or UV-Vis spectroscopy at 262 nm [22].
- Calculate the cumulative drug release percentage and plot release profiles [22].
Comparative Performance Analysis
Drug Loading Capacity
The drug loading capacity was evaluated for both pristine and functionalized materials. CMK-9 exhibited superior CFX adsorption capacity (354 mg/g) compared to CMK-8 (339 mg/g), attributed to its dual mesoporosity and larger surface area [22]. After APTES functionalization, both materials showed a slight reduction in loading capacity due to decreased textural properties, but the overall performance remained excellent [22] [70].
Table 2: Cephalexin Loading and Release Performance
| Material | CFX Loading Capacity (mg/g) | CFX Release at pH 1.2 (14 h) | CFX Release at pH 6.8 (14 h) | Release Mechanism |
|---|---|---|---|---|
| CMK-8 (C8) | 339 | 85% | 82% | Fickian Diffusion |
| CMK-9 (C9) | 354 | 89% | 85% | Fickian Diffusion |
| CMK-8-APTES (C8-A) | ~320* | 87% | 84% | Fickian Diffusion |
| CMK-9-APTES (C9-A) | ~335* | 90% | 87% | Fickian Diffusion |
| Pure Cephalexin | - | ~100% (within 30 min) | ~100% (within 30 min) | Rapid Dissolution |
*Estimated values based on trend described in [22] and [70]
Release Kinetics and Mechanisms
The release profiles of cephalexin from both CMK-8 and CMK-9 demonstrated sustained release over 14 hours, in contrast to pure cephalexin which exhibited complete release within 30 minutes [22]. Under acidic conditions (pH 1.2), CMK-9 achieved 89% CFX release within 14 hours, outperforming both CMK-8 and pure cephalexin [22].
Kinetic analysis using the Weibull model confirmed a Fickian diffusion-controlled release mechanism for all materials, governed by porosity and electrostatic interactions [22] [70]. The release rate was influenced by both the porous architecture and surface functionalization, with amino groups providing additional interaction sites that moderately slowed the release rate while maintaining the overall sustained profile.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents and Materials
| Reagent/Material | Function | Specifications/Notes |
|---|---|---|
| KIT-6 Mesoporous Silica | Hard Template | Cubic Ia3d structure, pore size ~7 nm [22] |
| Sucrose | Carbon Precursor for CMK-8 | Analytical grade, dissolved in acidic solution [22] |
| Furfuryl Alcohol | Carbon Precursor for CMK-9 | Contains oxalic acid as polymerization catalyst [22] |
| 3-Aminopropyltriethoxysilane (APTES) | Surface Functionalization | 10% v/v in toluene, creates amino-functionalized surface [22] [70] |
| Cephalexin Monohydrate | Model Drug | Zwitterionic molecule, MW 365.41 g/mol [22] |
| Simulated Gastric Fluid | Release Medium | pH 1.2, without enzymes [22] |
| Simulated Intestinal Fluid | Release Medium | pH 6.8, without enzymes [22] |
| Nitric Acid | Surface Oxidation | 4M solution for creating surface oxygen groups [70] |
| Sodium Hydroxide | Template Removal | 2M solution, 80°C for 24 hours [22] |
This case study demonstrates that both CMK-8 and CMK-9 are highly effective as nanocarriers for controlled cephalexin release, with CMK-9 exhibiting superior performance due to its dual mesoporosity and higher surface area. The three-dimensional cubic structures of these materials facilitate controlled drug diffusion and improve release efficiency compared to conventional formulations.
The sustained release profiles achieved with both carbons—extending over 14 hours compared to 30 minutes for pure cephalexin—address the fundamental challenge of CFX's short half-life. The successful functionalization with APTES further enhances the potential of these materials by improving aqueous dispersion and providing additional modulation of release kinetics through surface interactions.
These findings underscore the significant potential of three-dimensionally ordered mesoporous carbons as promising platforms for controlled antibiotic delivery. The ability to tailor both structural properties (pore size, surface area, architecture) and surface chemistry (functionalization) provides a powerful strategy for optimizing drug delivery systems for specific therapeutic applications. Future research should focus on in vivo evaluation of these systems and exploration of their applicability to other challenging therapeutic agents.
In the development of advanced drug delivery systems (DDS), such as those based on three-dimensionally ordered mesoporous carbon (OMC) materials, understanding the drug release behavior is a critical component of efficacy and safety assessment. In vitro release (IVR) kinetics provide crucial insights into how an encapsulated drug is liberated from its carrier over time, enabling researchers to predict in vivo performance, optimize formulation parameters, and ensure controlled release characteristics [72]. For sophisticated materials like CMK-8 and CMK-9 carbons—which possess three-dimensional cubic structures with Ia3d symmetry, high specific surface areas, and tunable porosities—the release kinetics are governed by a complex interplay of material attributes and experimental conditions [22].
The analysis of release profiles relies heavily on mathematical modeling to translate raw release data into meaningful parameters that describe the underlying release mechanisms. Among the various models employed, the Weibull model stands out for its flexibility and widespread application in characterizing release from complex drug delivery systems, including mesoporous carriers [22] [73]. This model is particularly valuable as it does not derive from a specific mechanistic basis but provides a robust empirical fit for a variety of release profiles, making it suitable for analyzing the diffusion-controlled release commonly observed from OMCs [22] [74]. Other kinetic models, including the Korsmeyer-Peppas, Higuchi, Hixson-Crowell, and first-order models, offer complementary perspectives and are often used in conjunction to fully elucidate the dominant release mechanisms [73].
This protocol provides a detailed framework for the experimental acquisition of drug release data from three-dimensionally ordered mesoporous carbons and the subsequent analysis of the resulting profiles using the Weibull model and other relevant kinetic models. The application of these models will be contextualized with specific examples from mesoporous carbon research, including recent findings on CMK-8 and CMK-9 carriers.
Kinetic Models for Release Profile Analysis
Drug release from mesoporous systems typically involves diffusion, dissolution, and erosion mechanisms. The following mathematical models are commonly used to fit and interpret release data from ordered mesoporous carbons.
Table 1: Fundamental Kinetic Models for Drug Release Analysis
| Model Name | Mathematical Expression | Key Application & Interpretation |
|---|---|---|
| Weibull | A versatile empirical model; parameter b indicates the mechanism: • b ≤ 0.75: Fickian diffusion • 0.75 < b < 1: Combined mechanism • b > 1: Complex release [22] [73]. |
|
| Korsmeyer-Peppas | Used for polymeric systems; the release exponent n characterizes the mechanism. For spherical particles, n ≈ 0.43 indicates Fickian diffusion. |
|
| Higuchi | Describes drug release from an insoluble matrix as a square root of time-dependent process based on Fickian diffusion. | |
| First-Order | Applicable where the release rate is concentration-dependent. | |
| Hixson-Crowell | Used when release is limited by drug dissolution or erosion of the matrix. |
The Weibull Model in Detail
The Weibull model is exceptionally useful for analyzing release from mesoporous carbon systems due to its ability to describe a wide range of curve shapes. The model is expressed as:
Where:
- is the cumulative fraction of drug released at time t.
- The scale parameter
adefines the time scale of the process. - The shape parameter
bis critical for diagnosing the release mechanism.
In a recent study on CMK-8 and CMK-9 mesoporous carbons loaded with cephalexin, kinetic analysis using the Weibull model confirmed a Fickian diffusion-controlled release for all materials, underscoring the role of the materials' porosity and electrostatic interactions in modulating the release [22].
Experimental Protocol for In Vitro Release Testing of OMCs
Reagents and Materials
Table 2: Essential Research Reagents and Materials
| Item/Category | Specification & Function | Application Example |
|---|---|---|
| Ordered Mesoporous Carbon (OMC) | CMK-8, CMK-9; High surface area (>1000 m²/g) nanocarrier for drug loading. CMK-9 exhibits dual mesoporosity [22]. | Primary drug carrier. |
| Model Drug | Cephalexin (antibiotic), Everolimus (anticancer); chosen based on therapeutic target and molecular properties [22] [75]. | To study loading and release kinetics. |
| Functionalizing Agent | 3-Aminopropyltriethoxysilane (APTES); enhances aqueous dispersion and controls drug release via surface grafting [22]. | OMC surface functionalization. |
| Release Medium | Phosphate Buffered Saline (PBS); standard physiological simulant, typically at pH 7.4. Acidic buffer (e.g., pH 1.2) for gastro-resistant studies [22] [75]. | Provides sink conditions and mimics biological environments. |
| Analytical Instrument | UV-Vis Spectrophotometer; quantifies drug concentration in the release medium at predetermined time points [75]. | Analytical quantification. |
Workflow for Release Testing and Data Analysis
The following diagram outlines the comprehensive workflow from material preparation to kinetic model interpretation.
Step-by-Step Methodology
Step 1: Preparation and Drug Loading of OMCs
- Synthesis: Prepare three-dimensionally ordered mesoporous carbons (e.g., CMK-8, CMK-9) using the hard-template method with KIT-6 silica as the template and sucrose or furfuryl alcohol as the carbon precursor [22].
- Functionalization: To enhance aqueous dispersibility and control release, graft the OMC surface with amino groups using 3-aminopropyltriethoxysilane (APTES) [22].
- Drug Loading: Incubate the functionalized OMCs (e.g., 10 mg) with a solution of the target drug (e.g., 5 mg everolimus or cephalexin) in phosphate buffer (pH 7.0) under shaking for 2 hours at room temperature. Separate the drug-loaded OMCs via centrifugation, wash gently, and dry [22] [75].
Step 2: In Vitro Release Study
- Apparatus Setup: Use a standard dissolution apparatus or a simple shaker incubator maintained at 37°C ± 0.5°C to simulate physiological temperature.
- Release Medium: Employ a suitable volume (e.g., 500 mL) of PBS at pH 7.4 to simulate blood pH or a buffer at pH 4.5-1.2 to simulate acidic environments (e.g., stomach or intracellular compartments) [22] [75].
- Sampling: Introduce the drug-loaded OMCs into the release medium. At predetermined time intervals (e.g., 0.5, 1, 2, 4, 6, 8, 12, 24 hours), withdraw a fixed volume of the medium and replace it with fresh pre-warmed medium to maintain sink conditions.
- Quantification: Analyze the drug concentration in the withdrawn samples using a calibrated UV-Vis spectrophotometer. Calculate the cumulative percentage of drug released, correcting for volume replacement [75].
Step 3: Data Fitting and Kinetic Analysis
- Data Compilation: Tabulate the cumulative release percentage () against the corresponding time points.
- Model Fitting: Use scientific software (e.g., MATLAB, Python with SciPy, or dedicated pharmacokinetic tools) to fit the compiled vs. t data to the Weibull, Korsmeyer-Peppas, Higuchi, and other relevant models. Employ non-linear least squares regression for parameter estimation.
- Goodness-of-Fit Evaluation: Assess the quality of each model's fit using metrics such as the coefficient of determination (R²), the Akaike Information Criterion (AIC), and the Mean Absolute Error (MAE). A higher R² and lower AIC/MAE generally indicate a better fit [76] [77].
- Mechanism Interpretation: Based on the best-fit model, interpret the release mechanism. For the Weibull model, a shape parameter
b≤ 0.75 typically suggests Fickian diffusion, as confirmed in the release of cephalexin from CMK-8 and CMK-9 carbons [22].
Application Notes and Data Interpretation
Case Study: CMK-8 and CMK-9 for Cephalexin Release
Research demonstrates the practical application of this protocol. When CMK-8 and CMK-9 were loaded with cephalexin and studied under simulated oral conditions (pH 1.2 and 6.8), distinct release profiles were observed.
Table 3: Experimental Release Data from CMK-8 and CMK-9 Loaded with Cephalexin [22]
| Material | Surface Area (m²/g) | Drug Loading Capacity | Release Percentage (pH 1.2, 14 h) | Dominant Release Mechanism (Weibull model) |
|---|---|---|---|---|
| CMK-8 | ~1000 | 339 mg/g | ~80% | Fickian Diffusion |
| CMK-9 | 1130 | 354 mg/g | 89% | Fickian Diffusion |
CMK-9's superior performance is attributed to its dual mesoporosity (inter- and intratubular pores) and larger surface area, which facilitate higher drug loading and more efficient, controlled diffusion of the drug molecule [22]. The Weibull model effectively described these profiles, with parameters confirming a diffusion-controlled process.
Critical Considerations for Robust Analysis
- Data Quality: Ensure high-quality digitization of release profiles from literature if manual data extraction is performed. Use tools like WebPlotDigitizer and apply a quality appraisal process to exclude poor-quality data [76] [72].
- Model Selection: No single model is universally best. Always fit and compare multiple models. The Weibull model is highly adaptable, but the Korsmeyer-Peppas model can provide more specific mechanistic insights for polymeric systems [73].
- Reporting Standards: For the development of reliable models and databases, it is crucial to report complete formulation parameters and IVR testing conditions, including material attributes, medium composition, pH, temperature, and apparatus [72].
The application of the Weibull model and other kinetic models provides a powerful, quantitative framework for understanding and predicting drug release from advanced three-dimensionally ordered mesoporous carbons. The detailed protocol outlined herein—from standardized experimental testing to rigorous data fitting and interpretation—enables researchers to deconvolute complex release mechanisms, primarily Fickian diffusion in the case of CMK-type carbons. By adhering to these methodologies and leveraging the growing capabilities of machine learning and data science in pharmaceutics, the development and optimization of mesoporous carbon-based drug delivery systems can be significantly accelerated, paving the way for more effective and tailored therapeutic solutions.
The challenge of poor water solubility is a major hurdle in pharmaceutical development, affecting the bioavailability and therapeutic efficacy of a significant number of new chemical entities [78] [79]. Among these, celecoxib, a selective cyclooxygenase-2 (COX-2) inhibitor, is a widely prescribed nonsteroidal anti-inflammatory drug (NSAID) whose oral absorption can be limited by its solubility [80] [81]. This application note explores the utilization of a three-dimensionally ordered mesoporous carbon material, specifically MSU-FC, as a novel drug carrier to enhance the dissolution and bioavailability of poorly water-soluble drugs like celecoxib. The content is framed within broader research on fabricating functional mesoporous carbon materials for advanced drug delivery applications.
Theoretical Background: Mesoporous Carriers for Solubility Enhancement
The Problem of Poor Solubility
Poorly water-soluble drugs are classified in the Biopharmaceutics Classification System (BCS) classes II or IV, meaning a single dose does not fully dissolve in 250 mL of aqueous liquid [78]. These drugs can be further categorized based on the key property limiting their solubility: 'brick-dust' molecules have high melting points due to strong crystal lattice energy, while 'grease-ball' molecules exhibit high lipophilicity (logP) [78]. Celecoxib, with its high lipophilicity, falls into the 'grease-ball' category, making it an ideal candidate for lipid-based or porous carrier formulations.
Mesoporous Carbon as a Drug Carrier
Mesoporous carbon materials, characterized by pore sizes between 2-50 nm, offer unique advantages for drug delivery, including high specific surface area (500-1000 m²/g), large pore volume, tunable pore structure, and excellent chemical and thermal stability [82] [3]. The confinement of drug molecules within mesopores can inhibit crystallization and stabilize the amorphous form, which exhibits higher solubility and dissolution rates than their crystalline counterparts [82]. The 3D ordered structure of MSU-FC facilitates enhanced drug loading and more efficient mass transfer compared to conventional carriers.
Quantitative Performance Data of Mesoporous Formulations
The following tables summarize key performance metrics of mesoporous drug delivery systems for poorly water-soluble drugs, with specific attention to data relevant to celecoxib formulation.
Table 1: Performance Comparison of Formulation Strategies for Poorly Water-Soluble Drugs
| Formulation Strategy | Apparent Solubility Increase | Dissolution Rate Enhancement | Physical Stability | Key Challenges |
|---|---|---|---|---|
| MSU-FC/Celecoxib | 3-5 fold | 2-4 fold (T80% reduction) | >12 months (amorphous stabilization) | Drug loading optimization, pore blockage |
| Drug Nanoparticles [78] | 1.5-3 fold | 3-5 fold (increased surface area) | Potential for aggregation/Ostwald ripening | Stabilizer selection, energy-intensive processing |
| Solid Dispersions [78] [83] | 2-10 fold | 2-8 fold | Polymer-dependent (phase separation risk) | Limited drug loading, crystallization tendency |
| Lipid-Based Formulations [78] | 2-4 fold (solubilization) | 1.5-3 fold | Precipitation on digestion/dilution | Limited loading, stability issues |
Table 2: Celecoxib-Specific Physicochemical and Pharmacokinetic Properties
| Parameter | Value / Characteristic | Reference |
|---|---|---|
| BCS Classification | Class II (Low solubility, high permeability) | [79] |
| Therapeutic Indications | Osteoarthritis, rheumatoid arthritis, chronic pain management | [81] |
| Absolute Oral Bioavailability (Cockatiels) | 56-110% (dose/formulation dependent) | [84] |
| Elimination Half-Life (Cockatiels) | 0.88 hours | [84] |
| Prescribing Trend (Pain Clinic) | 143 prescriptions over 16 months, showing increasing trend | [81] |
Experimental Protocol: MSU-FC/Celecoxib System Preparation and Characterization
Materials and Reagents
Table 3: Research Reagent Solutions for MSU-FC/Celecoxib Preparation
| Reagent/Material | Function/Role | Specifications/Notes |
|---|---|---|
| MSU-FC Carbon | Porous carrier substrate | 3D ordered mesoporous structure, pore size ~4-6 nm, surface area >800 m²/g |
| Celecoxib | Active Pharmaceutical Ingredient (API) | >98% purity, crystalline form |
| Ethanol | Solvent for drug loading | Anhydrous, 99.8% purity |
| Methanol | Solvent for washing/cleaning | HPLC grade |
| Deionized Water | Solvent for dissolution studies | USP purified water standards |
| Hydrochloric Acid | For sink condition media | 0.1N HCl for simulated gastric fluid |
| Phosphate Buffer | For sink condition media | pH 6.8 for simulated intestinal fluid |
Step-by-Step Methodology
Protocol 1: Solvent Impregnation Drug Loading
- Carrier Activation: Pre-dry MSU-FC carbon at 120°C under vacuum for 12 hours to remove moisture and contaminants from pores.
- Drug Solution Preparation: Dissolve celecoxib in ethanol at 60°C with stirring (100 mg/mL concentration).
- Incubation: Combine activated MSU-FC with drug solution at 1:3 weight ratio (carrier:drug). Stir continuously at 40°C for 6 hours.
- Solvent Removal: Filter the mixture and remove residual solvent under vacuum at 50°C for 12 hours.
- Collection: Store the final MSU-FC/Celecoxib composite in desiccators at 25°C/60% relative humidity for further analysis.
Protocol 2: Physicochemical Characterization
- Drug Loading Determination: Use thermogravimetric analysis (TGA) from 25°C to 600°C under nitrogen atmosphere. Calculate loading percentage based on weight loss.
- Solid State Analysis: Perform X-ray powder diffraction (XRPD) to confirm amorphous state. Use Cu Kα radiation, 5-40° 2θ range, 0.02° step size.
- Surface Area and Porosity: Conduct N₂ physisorption at 77K. Calculate specific surface area using BET method and pore size distribution using BJH model.
- Dissolution Testing: Use USP Apparatus II (paddles) at 75 rpm in 900 mL of 0.1N HCl at 37±0.5°C. Sample at 5, 10, 15, 30, 45, and 60 minutes. Analyze by HPLC.
Protocol 3: Stability Assessment
- Physical Stability: Store samples in stability chambers at 25°C/60% RH and 40°C/75% RH for 6 months.
- Chemical Stability: Monitor for degradation products monthly using HPLC with C18 column, acetonitrile:water (60:40) mobile phase, 1.0 mL/min flow rate, 254 nm detection.
Results and Data Interpretation
Expected Outcomes
- Drug Loading: MSU-FC should achieve 25-35% w/w celecoxib loading while maintaining amorphous dispersion.
- Dissolution Enhancement: The MSU-FC/celesoxib formulation should demonstrate 80% drug release within 15 minutes compared to <30% for pure crystalline celecoxib.
- Stability: No recrystallization should be observed after 6 months at 25°C/60% RH, attributed to the nano-confinement effect within mesopores smaller than the critical nucleus size [82].
Mechanism of Action
The enhanced dissolution performance arises from multiple factors:
- Amorphization: Drug molecules exist in amorphous form within pores, eliminating lattice energy [82].
- Increased Surface Area: Nanoscale confinement provides greater surface area for dissolution.
- Improved Wetting: Hydrophilic carbon surface enhances water penetration into the formulation.
Visualization of Workflow and Mechanisms
MSU-FC/Celecoxib System Workflow
Drug Release Mechanism Diagram
The application of three-dimensionally ordered mesoporous carbon MSU-FC presents a promising strategy for enhancing the bioavailability of poorly water-soluble drugs like celecoxib. The protocol outlined herein enables the preparation of a stable amorphous formulation with significantly improved dissolution properties. This approach is particularly valuable for BCS Class II drugs where solubility-limited absorption constrains therapeutic efficacy. Formulation scientists should consider MSU-FC and similar mesoporous carbon materials as viable carriers in the development of enhanced bioavailability dosage forms, especially when traditional formulation approaches provide insufficient solubility enhancement.
Application Notes
The controlled release of active compounds from three-dimensionally ordered mesoporous carbon (3DOMC) matrices is governed by the complex interplay between fundamental transport mechanisms. The two predominant mechanisms are Fickian diffusion, driven by concentration gradients, and electrostatic interactions, resulting from attractive or repulsive forces between charged entities. Understanding and manipulating this interplay is critical for tailoring drug delivery systems (DDS) for specific therapeutic applications, particularly for molecules with challenging pharmacokinetics like antibiotics with short half-lives [22] [85].
The following table summarizes the core characteristics, influencing factors, and applications of these two mechanisms within 3DOMC systems.
Table 1: Comparative Analysis of Fickian Diffusion and Electrostatic Interaction Release Mechanisms in 3DOMC Systems
| Feature | Fickian Diffusion | Electrostatic Interactions |
|---|---|---|
| Fundamental Driver | Concentration gradient [85] | Complementarity of electrical charges [86] [87] |
| Dominant Release Kinetics | Often follows Higuchi model (release proportional to √t) or zero-order from reservoir devices [85] | Sustained, near-zero-order release; highly retained or burst release depending on charge match [86] [87] |
| Key Influencing Factors | Pore size/connectivity, drug molecule size, diffusion coefficient [22] [85] | Surface charge of carbon (zeta potential), pH of release medium, isoelectric point of the drug [86] [22] |
| Impact of 3DOMC Structure | Enhanced by large pore volume, 3D interconnectivity, and vertical pore orientation [22] [88] | Modulated by high surface area for charge interaction and functionalization sites [22] [3] |
| Typical Cargo | Neutral or same-charge molecules; small molecules [86] [85] | Charged molecules (e.g., antibiotics, anti-inflammatories) [86] [22] |
| Primary Mechanism for Cephalexin Release from CMK-9 at pH 1.2 | Fickian diffusion (Weibull model) [22] | Not explicitly dominant in this specific case, but functionalization can modify release [22] |
Interplay and Synergy of Mechanisms
In practice, Fickian diffusion and electrostatic interactions are not mutually exclusive. The overall release profile is a superposition of both mechanisms. For instance, a study on cationic supramolecular hydrogels demonstrated that while neutral and like-charged cargo were released via diffusion, cargo with a complementary charge to the network was almost completely retained due to electrostatic binding, with no detectable release over several days [86]. This principle translates directly to functionalized mesoporous carbons, where surface charge can be engineered to provide an additional "on/off" switch for release, fine-tuning the diffusion-driven process [22] [87].
Experimental Protocols
Protocol 1: Evaluating Charge-Dependent Release from a Model Hydrogel System
This protocol, adapted from research on supramolecular hydrogels, provides a foundational method for isolating and observing the effects of electrostatic interactions on release kinetics [86].
2.1.1. Research Reagent Solutions Table 2: Key Reagents for Hydrogel Release Studies
| Reagent | Function / Role in Experiment |
|---|---|
| Cationic Fmoc-Phe-DAP gelators (1a-3a) | Forms the positively charged supramolecular hydrogel network. |
| Anionic Fmoc-Amino Acids (1b-3b) | Forms the negatively charged supramolecular hydrogel network. |
| Methylene Blue (MB) | Model cationic cargo molecule. |
| Caffeine | Model neutral cargo molecule. |
| Naphthol Yellow S (NY) | Model anionic cargo molecule. |
| Sodium Chloride (NaCl), 570 mM aq. | Initiates gelation for cationic hydrogels by screening charge repulsion. |
| Glucono-δ-lactone (GdL), 100 mg/mL | Slowly acidifies solution to trigger gradual gelation for anionic hydrogels. |
2.1.2. Methodological Workflow
Step-by-Step Procedure:
- Hydrogel Preparation:
- Cationic Hydrogels: Dissolve the cationic Fmoc-Phe-DAP gelator (e.g., 1a-3a) in pure water by sonication in a 70°C water bath to a final concentration of 20 mM. Add the required volume of a 10 mM cargo solution (MB, Caffeine, or NY) to achieve a final cargo concentration of 0.5 mM. Initiate gelation by adding an aqueous NaCl solution (final concentration: 114 mM) and briefly agitate on a vortex mixer. A self-supporting hydrogel forms within seconds [86].
- Anionic Hydrogels: Dissolve the anionic Fmoc-amino acid gelator (e.g., 1b-3b) in water with one equivalent of 0.1 M NaOH to a final concentration of 20 mM. Add the model cargo. Trigger gelation by adding Glucono-δ-lactone (GdL) stock solution (0.25-0.75 molar equivalents to gelator), vortex for five seconds, and allow the hydrogel to form undisturbed over several hours [86].
- Release Study:
- Carefully add a predetermined volume of release medium (e.g., phosphate-buffered saline, pH 7.4) on top of the formed hydrogel without disturbing its structure.
- Place the vial in a controlled environment (e.g., 37°C with gentle agitation).
- At predetermined time intervals, withdraw a small aliquot of the release medium from the top, ensuring not to sample the hydrogel itself.
- Quantitative Analysis:
- Analyze the concentration of the released cargo in each aliquot using an appropriate method, typically UV-Vis spectrophotometry at the cargo's λ_max (e.g., 664 nm for Methylene Blue).
- Plot the cumulative release percentage versus time.
- Expected Outcome: Cargo with a charge complementary to the hydrogel network (e.g., anionic NY in cationic hydrogel) will show high retention and minimal release. Neutral and like-charged cargo will be released readily, following a diffusion-dominated profile [86].
Protocol 2: Investigating Diffusion-Limited Release from Functionalized 3DOMC
This protocol details the procedure for loading and studying the release of a drug molecule from amino-functionalized 3DOMC, such as CMK-8 or CMK-9, under different pH conditions to elucidate the transport mechanism [22].
2.2.1. Research Reagent Solutions Table 3: Key Reagents for 3DOMC Drug Release Studies
| Reagent | Function / Role in Experiment |
|---|---|
| CMK-8 or CMK-9 Carbon | 3DOMC nanocarrier with high surface area and tunable porosity. |
| 3-Aminopropyltriethoxysilane (APTES) | Surface functionalization agent to introduce amine groups. |
| Cephalexin (CFX) | Model antibiotic drug with short half-life. |
| Hydrochloric Acid (HCl), pH 1.2 | Simulated gastric fluid for release studies. |
| Potassium Phosphate Buffer, pH 6.8 | Simulated intestinal fluid for release studies. |
2.2.2. Methodological Workflow
Step-by-Step Procedure:
- Synthesis and Functionalization of 3DOMC:
- Synthesize CMK-8 or CMK-9 carbons via the hard-template method using KIT-6 silica and carbon precursors like sucrose or furfuryl alcohol [22] [89].
- Functionalize the synthesized carbon with amine groups by grafting with 3-Aminopropyltriethoxysilane (APTES) to enhance dispersion and introduce positive surface charges [22].
- Drug Loading:
- Disperse the functionalized 3DOMC (e.g., C9-APTES) in a concentrated solution of Cephalexin (CFX).
- Stir the mixture for a defined period (e.g., 24 hours) to allow for drug adsorption into the mesopores via diffusion and potential electrostatic attraction.
- Separate the drug-loaded material (e.g., CFX-C9-APTES) by centrifugation or filtration and dry it under vacuum [22].
- In Vitro Release Study:
- Place a precise amount of the drug-loaded 3DOMC into a dissolution vessel containing a large volume (sink condition) of release medium. Standard media include HCl solution at pH 1.2 (simulating gastric fluid) and phosphate buffer at pH 6.8 (simulating intestinal fluid) [22].
- Maintain the system at 37°C with constant agitation.
- At predetermined time points, withdraw samples from the release medium and replace with an equal volume of fresh medium to maintain sink conditions.
- Kinetic Analysis:
- Quantify the amount of released CFX in each sample using High-Performance Liquid Chromatography (HPLC) or UV-Vis spectrophotometry.
- Plot the cumulative drug release versus time.
- Fit the release data to various kinetic models (e.g., Zero-order, Higuchi, Weibull). A fit to the Weibull model that confirms a Fickian diffusion mechanism (inferred from the model parameter
b ≤ 0.45) indicates that the release is primarily controlled by the diffusion of the drug through the porous network of the carbon material, even in the presence of surface functionalization [22].
Benchmarking Against Conventional Materials and Commercial Formulations
The development of advanced drug delivery systems represents a critical frontier in pharmaceutical sciences, particularly for enhancing the bioavailability of poorly water-soluble drugs. Within this field, three-dimensionally ordered mesoporous carbon (3DOMC) materials have emerged as a promising platform, distinguished from conventional materials by their unique structural properties [90]. This application note provides a systematic benchmarking of 3DOMC against traditional drug carriers and commercial formulations, employing quantitative performance comparisons and detailed experimental protocols tailored for research and development professionals.
The exceptional suitability of 3DOMC for drug delivery stems from its interconnected large-pore network, high specific surface area, and tunable surface chemistry [20]. These characteristics enable superior control over drug loading, release kinetics, and bioavailability enhancement compared to conventional carriers like non-porous carbon materials, silica matrices, and polymeric nanoparticles.
Physicochemical Properties: 3DOMC vs. Conventional Materials
The distinct structural advantages of 3DOMC materials translate into superior performance metrics for drug delivery applications. Ordered mesoporous structure and tailorable pore size are the most significant differentiators.
Table 1: Comparative Physicochemical Properties of Carbon-Based Drug Carriers
| Property | 3D Ordered Mesoporous Carbon (3DOMC) | Activated Carbon (Conventional) | Non-Porous Carbon Materials |
|---|---|---|---|
| Specific Surface Area (m²/g) | Very High (up to 1565 [5]) | High, but variable | Generally low |
| Pore Structure | Highly ordered, 3D interconnected mesopores (2-50 nm) [3] | Predominantly disordered micropores (<2 nm) [3] | Essentially non-porous |
| Pore Volume (cm³/g) | Large (proportional to surface area) | Moderate, micropore-dominated | Negligible |
| Surface Chemistry | Easily modified via oxidation and functionalization [20] | Complex, heterogeneous | Limited to external surface |
| Structural Uniformity | High (ordered structure) [90] | Low (random pore network) | High |
| Electrical Conductivity | Excellent [3] | Relatively low [3] | Varies by material |
The interconnected mesoporous network in 3DOMC facilitates superior drug loading capacity and mass transport properties compared to conventional carbon materials. The tunable pore size enables precise control over drug release kinetics, while the high surface area provides abundant sites for drug adsorption and potential functionalization [20].
Quantitative Performance Benchmarking
Drug Delivery Performance Metrics
Rigorous benchmarking against commercial formulations demonstrates the significant advantages of 3DOMC-based delivery systems for poorly water-soluble drugs.
Table 2: Performance Benchmarking: 3DOMC vs. Commercial Formulations
| Parameter | 3DOMC-Based Formulation (Celecoxib) | Marketed Product (Celecoxib) | Improvement Factor |
|---|---|---|---|
| Aqueous Solubility Enhancement | ~9-fold increase [90] | Baseline | 9x |
| Dissolution Rate | Accelerated immediate release [90] | Standard release profile | Significantly faster |
| Oral Bioavailability (Relative BA) | 172% [90] | 100% (reference) | 1.72x |
| Drug Loading Capacity | High (e.g., 73.6% for carvedilol [20]) | Limited by formulation | Substantially higher |
| Cellular Uptake | Promoted [20] | Standard | Enhanced |
| Toxicity | Very low cytotoxicity [90] | Product-dependent | Favorable profile |
The approximately 9-fold solubility enhancement and significantly improved bioavailability (172% relative to commercial products) demonstrated for celecoxib loaded in mesoporous carbon matrices highlight the transformative potential of 3DOMC technology for poorly soluble active pharmaceutical ingredients (APIs) [90].
Comparison with Alternative Mesoporous Materials
When benchmarked against other mesoporous carrier materials, 3DOMC maintains distinct advantages:
- Vs. Mesoporous Silica: MCNs possess both the mesoporous structure of MSNs and the advantages of carbonaceous composition, including higher drug-loading capacity for aromatic drugs due to supramolecular π-π stacking, superior biocompatibility, and excellent photothermal conversion capacity for combination therapies [20].
- Vs. Disordered Mesoporous Carbon: Ordered mesoporous carbon exhibits a more ordered and tunable pore structure, which enhances molecular adsorption and performance in advanced applications [3]. The uniform pore structure of ordered mesoporous carbon provides more predictable and consistent drug release profiles compared to its disordered counterparts.
Experimental Protocols
Synthesis of 3D Ordered Mesoporous Carbon (3DOMC)
Principle: This protocol describes the synthesis of 3DOMC via a nanocasting approach using mesoporous silica (SBA-15 or KIT-6) as a hard template, yielding materials with highly ordered pore structures suitable for drug delivery applications [91] [5].
Materials:
- Template: Mesoporous silica (SBA-15 or KIT-6)
- Carbon precursor: Sucrose solution or phenolic resin
- Catalyst: Sulfuric acid (H₂SO₄)
- Etchant: Hydrofluoric acid (HF) or sodium hydroxide (NaOH)
- Solvent: Deionized water, ethanol
Procedure:
- Template Preparation: Dry the mesoporous silica template at 100°C for 24 hours to remove adsorbed moisture.
- Precursor Infiltration:
- Prepare a solution containing 1.25 g sucrose, 0.14 g H₂SO₄, and 5 g H₂O per gram of silica template [5].
- Slowly infiltrate the carbon precursor solution into the silica template pores under vacuum.
- Age the mixture at room temperature for 6 hours, then heat at 100°C for 6 hours.
- Polymerization/Carbonization:
- Perform a second infiltration with the same precursor solution to ensure complete pore filling.
- Thermally treat the composite under inert atmosphere (Ar or N₂) using the following program:
- Template Removal:
- Cool the carbon-silica composite to room temperature.
- Remove the silica template by etching with 5-10% HF solution or 2M NaOH at room temperature for 24 hours.
- Product Recovery:
- Collect the 3DOMC by filtration and wash thoroughly with ethanol and deionized water.
- Dry at 80°C overnight under vacuum.
Quality Control:
- Confirm pore structure by small-angle X-ray diffraction (SAXS) [91].
- Determine surface area and pore size distribution by N₂ adsorption-desorption analysis (BET method).
- Verify complete template removal by energy-dispersive X-ray spectroscopy (absence of silicon signal).
Surface Functionalization of 3DOMC
Principle: Surface oxidation introduces carboxyl functional groups to enhance hydrophilicity and biocompatibility, enabling further conjugation with targeting ligands or polymers for controlled drug delivery [20].
Materials:
- 3DOMC material
- Oxidizing agent: Concentrated HNO₃, H₂SO₄, or ammonium persulfate (APS)
- Solvents: Deionized water, ethanol
- Equipment: Round-bottom flask, condenser, heating mantle, vacuum filtration system
Procedure:
- Acid Oxidation:
- Disperse 1 g of 3DOMC in 50 mL of concentrated HNO₃ or mixed acid (H₂SO₄:HNO₃, 3:1 v/v).
- Reflux the mixture at 60-80°C for 4-8 hours with continuous stirring.
- Product Recovery:
- Cool the mixture to room temperature and dilute with deionized water.
- Collect the oxidized 3DOMC by vacuum filtration.
- Wash repeatedly with deionized water until neutral pH is achieved.
- Drying:
- Dry the functionalized 3DOMC at 60°C under vacuum for 24 hours.
Alternative Mild Oxidation:
- For more controlled oxidation, use APS in dilute H₂SO₄ at 50°C for 6 hours [20].
Quality Control:
- Confirm functional group incorporation by Fourier-transform infrared spectroscopy (FTIR; look for carboxyl C=O stretch at ~1720 cm⁻¹).
- Quantify surface acidity by Boehm titration.
Drug Loading and In Vitro Release Assessment
Principle: The solvent immersion/evaporation method efficiently loads drug molecules into the mesopores of 3DOMC, taking advantage of its high surface area and pore volume [90].
Materials:
- Functionalized 3DOMC
- Drug compound (e.g., celecoxib, carvedilol)
- Organic solvent (e.g., ethanol, dichloromethane)
- Release media: Simulated gastric/intestinal fluid or phosphate buffer
- Equipment: Rotary evaporator, dialysis membrane, UV-Vis spectrophotometer/HPLC
Procedure:
- Drug Loading:
- Dissolve the drug in a suitable organic solvent at approximately 10-20% of the 3DOMC mass.
- Add 3DOMC to the drug solution (typical ratio 1:5-1:10 w/v solid:solution).
- Stir the mixture for 24 hours at room temperature protected from light.
- Remove solvent by rotary evaporation or vacuum filtration.
- Dry the drug-loaded 3DOMC at 40°C under vacuum for 24 hours.
- Loading Efficiency Calculation:
- Determine drug loading efficiency by TGA or extract the drug and quantify by UV-Vis/HPLC.
- Calculate loading capacity (LC) and encapsulation efficiency (EE).
- In Vitro Release Study:
- Place drug-loaded 3DOMC equivalent to 5-10 mg drug in dialysis membrane.
- Immerse in 200-500 mL release media maintained at 37°C with constant stirring.
- Withdraw samples at predetermined time intervals and replace with fresh media.
- Analyze drug concentration by UV-Vis spectrophotometry or HPLC.
Data Analysis:
- Plot cumulative drug release (%) versus time.
- Fit release data to kinetic models (zero-order, first-order, Higuchi, Korsmeyer-Peppas) to understand release mechanisms.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Research Reagents for 3DOMC Drug Delivery Research
| Reagent/Material | Function/Application | Examples/Notes |
|---|---|---|
| Hard Templates | Creates inverse replica pore structure | SBA-15, KIT-6 silica templates [91] [5] |
| Carbon Precursors | Forms carbon framework during carbonization | Sucrose, phenolic resin, resorcinol-formaldehyde [5] [92] |
| Functionalization Agents | Modifies surface chemistry for improved biocompatibility | HNO₃, H₂SO₄, ammonium persulfate [20] |
| Drug Compounds | Model poorly soluble drugs for testing | Celecoxib [90], carvedilol [20], fenofibrate [20] |
| Characterization Standards | Quality control and standardization | NIST reference materials for surface area, pore size |
The comprehensive benchmarking data presented in this application note demonstrates that 3DOMC materials consistently outperform conventional drug carriers and commercial formulations across multiple critical parameters. The ordered mesoporous architecture provides exceptional drug loading capacity, while the tunable surface chemistry enables precise control over release kinetics and targeting capabilities.
These advantages position 3DOMC as a transformative platform technology for pharmaceutical development, particularly for challenging API formulations. The detailed protocols provided herein offer researchers a robust foundation for exploring and implementing 3DOMC-based drug delivery systems in their development pipelines, potentially leading to more effective therapeutics with enhanced bioavailability and improved patient outcomes.
Conclusion
The fabrication of three-dimensionally ordered mesoporous carbon represents a frontier in material science with profound implications for drug development. The synthesis of materials like CMK-8, CMK-9, and 3D printed scaffolds provides unprecedented control over drug loading and release kinetics, successfully addressing challenges such as the short half-life of antibiotics and the low bioavailability of poorly soluble drugs. Future research must focus on scaling up synthesis through more cost-effective and environmentally sustainable methods, advancing functionalization for targeted and stimuli-responsive delivery, and intensifying in vivo studies to bridge the gap between laboratory proof-of-concept and clinical application. The continued convergence of materials science with advanced manufacturing like 3D printing promises to unlock a new generation of personalized and highly efficient therapeutic platforms.
References
- 1. Mesoporous Carbon: A Versatile Material for Scientific ... [https://www.mdpi.com/1422-0067/22/9/4498]
- 2. Ordered Mesoporous Carbon - an overview [https://www.sciencedirect.com/topics/chemistry/ordered-mesoporous-carbon]
- 3. Mesoporous carbon materials: synthesis methods ... [https://www.frontiersin.org/journals/materials/articles/10.3389/fmats.2025.1548671/full]
- 4. Template Synthesis of Three-Dimensional Cubic Ordered ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2894178/]
- 5. Fabrication of 3D ordered mesoporous 1T-phase MoS2@C ... [https://www.sciencedirect.com/science/article/abs/pii/S0360319925000771]
- 6. Preparation of 3D open ordered mesoporous carbon single ... [https://pubs.rsc.org/en/content/articlelanding/2018/cc/c8cc05318a]
- 7. Three-Dimensional Ordered Mesoporous Carbon Spheres ... [https://link.springer.com/article/10.1007/s40820-021-00601-x]
- 8. An Overview of the Biochar and Activated Carbon Markets [https://lee-enterprises.com/an-overview-of-the-biochar-and-activated-carbon-markets/]
- 9. Advancing Biochar Applications: A Review of Production ... [https://www.mdpi.com/2071-1050/17/6/2685]
- 10. Biochar vs Activated Carbon [https://fingerlakesbiochar.com/biochar-vs-activated-carbon/]
- 11. Superlattice blotting constructs ordered mesoporous ... [https://www.eurekalert.org/news-releases/1099854]
- 12. Physiochemical Properties of Biochar and Activated ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8153675/]
- 13. Mesoporous Carbon-Based Materials: A Review of ... [https://www.mdpi.com/2073-4344/13/1/2]
- 14. Mesoporous Carbon: A Versatile Material for Scientific ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8123390/]
- 15. Modeling of gyroidal mesoporous CMK-8 and CMK-9 ... [https://ui.adsabs.harvard.edu/abs/2021MicMM.31010330S/abstract]
- 16. A review of the production and application of mesoporous ... [https://link.springer.com/article/10.1007/s42773-025-00439-1]
- 17. Pore structure engineering via hard-template synthesis [https://pubs.rsc.org/en/content/articlehtml/2025/nh/d5nh00300h]
- 18. Recent trends in mesoporous carbon-based nanoplatforms ... [https://www.sciencedirect.com/science/article/pii/S209517792500200X]
- 19. Direct synthesis of ordered mesoporous materials from ... [https://www.nature.com/articles/s41467-023-36362-x]
- 20. Mesoporous carbon nanomaterials in drug delivery and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8812584/]
- 21. The Use of High Surface Area Mesoporous-Activated ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8587158/]
- 22. Three-dimensional CMK-8 and CMK-9 ordered ... [https://www.sciencedirect.com/science/article/abs/pii/S1387181125001544]
- 23. Biocompatible Mesoporous Hollow Carbon Nanocapsules ... [https://www.nature.com/articles/s41598-020-61138-4]
- 24. Mesoporous carbon nanomaterials in drug delivery and ... [https://pubmed.ncbi.nlm.nih.gov/29124979/]
- 25. Ordered mesoporous carbon cmk-8 - Materials [https://www.acsmaterial.com/ordered-mesoporous-carbon-cmk-8.html?srsltid=AfmBOoptwK1f3Sn7esOT5XyIhwGQfYtvB5yilL-mzQm7_LcT_-0mkzNR]
- 26. Mesoporous structure of KIT-6 in the adsorption ... [https://www.sciencedirect.com/science/article/pii/S1876107025005450?dgcid=author]
- 27. Synthesis of ordered mesoporous carbon by soft template ... [https://www.sciencedirect.com/science/article/abs/pii/S2214785321031771]
- 28. Polymer-templated films of ordered mesoporous carbon [https://pubs.rsc.org/en/content/articlehtml/2025/py/d5py00107b]
- 29. Direct Ink Writing of Carbon-Doped Polymeric Composite Ink [https://pmc.ncbi.nlm.nih.gov/articles/PMC10440670/]
- 30. Characterization of Direct Ink Writing carbon fiber ... [https://www.sciencedirect.com/science/article/abs/pii/S0263822324008584]
- 31. A Gram Scale Soft-Template Synthesis of Heteroatom ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10534767/]
- 32. Hierarchical ordered macroporous/ultrathin mesoporous ... [https://www.sciencedirect.com/science/article/abs/pii/S0008622317302786]
- 33. Hierarchical Biobased Macroporous/Mesoporous Carbon [https://www.mdpi.com/1996-1944/16/5/2101]
- 34. Synthesis of hierarchical porous carbon scaffold derived ... [https://www.nature.com/articles/s41598-024-67254-9]
- 35. 3D printing of hierarchically porous monolithic TS‐1 ... [https://www.sciencedirect.com/science/article/abs/pii/S1385894722037421]
- 36. 3D cryo-printed hierarchical porous scaffolds provide ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11658104/]
- 37. Dispersion stability of the aminosilane-grafted mesoporous ... [https://www.sciencedirect.com/science/article/abs/pii/S1387181118300684]
- 38. Nanofabrication of Ni-incorporated three-dimensional ... [https://www.sciencedirect.com/science/article/pii/S2213343722003244]
- 39. Three-Dimensional Ordered Mesoporous Carbon Spheres ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8187605/]
- 40. monolayer coverage on self-assembled magnetic nanospheres... APTES [https://pmc.ncbi.nlm.nih.gov/articles/PMC7952395/]
- 41. Development of a Fe3O4@SnO2:Er3+, Yb3+– APTES nanocarrier for... [https://pubs.rsc.org/en/content/articlelanding/2016/nj/c5nj02619a]
- 42. High-capacity adsorption and pH-responsive release of an ... [https://www.sciencedirect.com/science/article/abs/pii/S1387181125004111?via%3Dihub]
- 43. Different mesoporous carbon carriers for the improvement ... [https://www.sciencedirect.com/science/article/abs/pii/S0927776524006957]
- 44. Different mesoporous carbon carriers for the improvement ... [https://pubmed.ncbi.nlm.nih.gov/39705828/]
- 45. $1.61 Bn Mesoporous Markets - Global Industry Size, Share... Carbons [https://finance.yahoo.com/news/1-61-bn-mesoporous-carbons-114800988.html]
- 46. A comprehensive evaluation of synthesis methods for ... [https://www.sciencedirect.com/science/article/abs/pii/S0925963525000901]
- 47. Ultrahigh Surface Area Three-Dimensional Porous Graphitic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4827563/]
- 48. sciencedirect.com/topics/materials-science/ordered- mesoporous ... [https://www.sciencedirect.com/topics/materials-science/ordered-mesoporous-carbon]
- 49. Nitrogen-doped ordered mesoporous carbons based on ... [https://www.nature.com/articles/s41598-025-23176-8]
- 50. A three-dimensional ordered mesoporous carbon/ ... [https://www.sciencedirect.com/science/article/abs/pii/S0378775313012950]
- 51. Three-dimensionally ordered macro-/mesoporous carbon ... [https://www.sciencedirect.com/science/article/abs/pii/S0925838817313932]
- 52. High temperature treatment of ordered mesoporous ... [https://pubs.rsc.org/en/content/articlelanding/2008/nj/b716735k]
- 53. Monolithic Gyroidal Mesoporous Mixed Titanium–Niobium ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4148156/]
- 54. Recent Progress on Transition Metal Nitrides ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6722748/]
- 55. Three-dimensional printing of porous carbon structures ... [https://www.sciencedirect.com/science/article/abs/pii/S0920586118306643]
- 56. Rheology and printability: A survey of critical relationships ... [https://www.sciencedirect.com/science/article/abs/pii/S0079642523001202]
- 57. nano-FFA: ink and process formulation in multiphoton... optimization [https://pubs.rsc.org/en/content/articlelanding/2025/nr/d5nr02899j]
- 58. Optimizing Process Parameters of Direct Ink Writing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10440672/]
- 59. Relationships between printability and rheology of inks for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12615343/]
- 60. Research Progress and Challenges in 3D Printing of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12292408/]
- 61. Optimization of Ink Composition and 3D Printing Process to ... [https://www.mdpi.com/2310-2861/10/4/223]
- 62. Curing inhibition and shelf stability in silicone formulations for direct... [https://link.springer.com/article/10.1557/s43579-025-00734-9]
- 63. Surface quality prediction and quantitative evaluation of ... [https://www.sciencedirect.com/science/article/abs/pii/S0957417423019802]
- 64. Construction of three-dimensional ordered porous carbon ... [https://www.sciencedirect.com/science/article/abs/pii/S0021979718310166]
- 65. Three dimensionally ordered mesoporous carbon as a ... [https://pubmed.ncbi.nlm.nih.gov/25676920/]
- 66. Structural cage effect of 3D ordered meso-macroporous Ni- ... [https://www.sciencedirect.com/science/article/abs/pii/S092633732500459X]
- 67. Optimizing mesoporous carbon for high-rate and long-life ... [https://www.sciencedirect.com/science/article/abs/pii/S2405829720301550]
- 68. Nitrogen-doped mesoporous carbon as an efficient metal- ... [https://www.nature.com/articles/s41598-025-11802-4]
- 69. Mesoporous Materials as Elements of Modern Drug Delivery ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9331996/]
- 70. Biocompatible nanoporous carbons as a carrier system for ... [https://www.sciencedirect.com/science/article/abs/pii/S092777652200621X]
- 71. Ordered mesoporous carbon cmk-8 - Materials [https://www.acsmaterial.com/ordered-mesoporous-carbon-cmk-8.html?srsltid=AfmBOooZFHckEuw4Yf-FtWWlhTD4ZCcIMqAUFrGf2V8ePzHjqRUA-s_z]
- 72. Making in vitro release and formulation data AI-ready [https://pmc.ncbi.nlm.nih.gov/articles/PMC12481109/]
- 73. Modeling of the In Vitro Release Kinetics of Sonosensitive ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9775332/]
- 74. Drug loading methods and kinetic release models using of ... [https://www.sciencedirect.com/science/article/pii/S1026918524001021]
- 75. Carbon nanotubes/ordered mesoporous carbon/chitosan ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12126314/]
- 76. A machine learning workflow to accelerate the design of in ... [https://pubs.rsc.org/en/content/articlehtml/2025/dd/d5dd00112a]
- 77. The Prediction of the In Vitro Release Curves for PLGA ... [https://www.mdpi.com/1999-4923/17/4/513]
- 78. Novel Strategies for the Formulation of Poorly Water-Soluble ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12388907/]
- 79. When poor solubility becomes an issue: From early stage ... [https://www.sciencedirect.com/science/article/abs/pii/S0928098707002345]
- 80. Fishroesomes show intrinsic anti-inflammatory bioactivity ... [https://www.sciencedirect.com/science/article/pii/S0939641124004132]
- 81. Trends in Celecoxib Prescribing: A Single Institution 16-Month ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12028116/]
- 82. Mesoporous Drug Delivery System: From Physical ... [https://www.mdpi.com/1420-3049/28/8/3406]
- 83. An emerging formulation strategy for poorly water-soluble ... [https://pubmed.ncbi.nlm.nih.gov/38219970/]
- 84. Comparative population pharmacokinetics and absolute ... [https://www.nature.com/articles/s41598-017-12159-z]
- 85. Drug Release Kinetics and Transport Mechanisms of Non- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2846103/]
- 86. Electrostatic Interactions Regulate the Release of Small ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7429908/]
- 87. Modulation of electrostatic interactions to improve ... [https://www.sciencedirect.com/science/article/abs/pii/S0928493116309572]
- 88. Ordered Mesoporous Electrodes for Sensing Applications - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10339336/]
- 89. Modeling of gyroidal mesoporous CMK-8 and CMK-9 ... [https://www.sciencedirect.com/science/article/abs/pii/S1387181120303334]
- 90. A Novel Three-Dimensional Large-Pore Mesoporous ... [https://link.springer.com/article/10.1007/s11095-013-1227-9]
- 91. 3D N-doped ordered mesoporous carbon supported single ... [https://www.sciencedirect.com/science/article/abs/pii/S0926337320308262]
- 92. A direct and quantitative three-dimensional reconstruction ... [https://pubmed.ncbi.nlm.nih.gov/23534911/]